Unscrambled eggs
A scientist’s eye for detail catches mixed-up frog cells reassembling
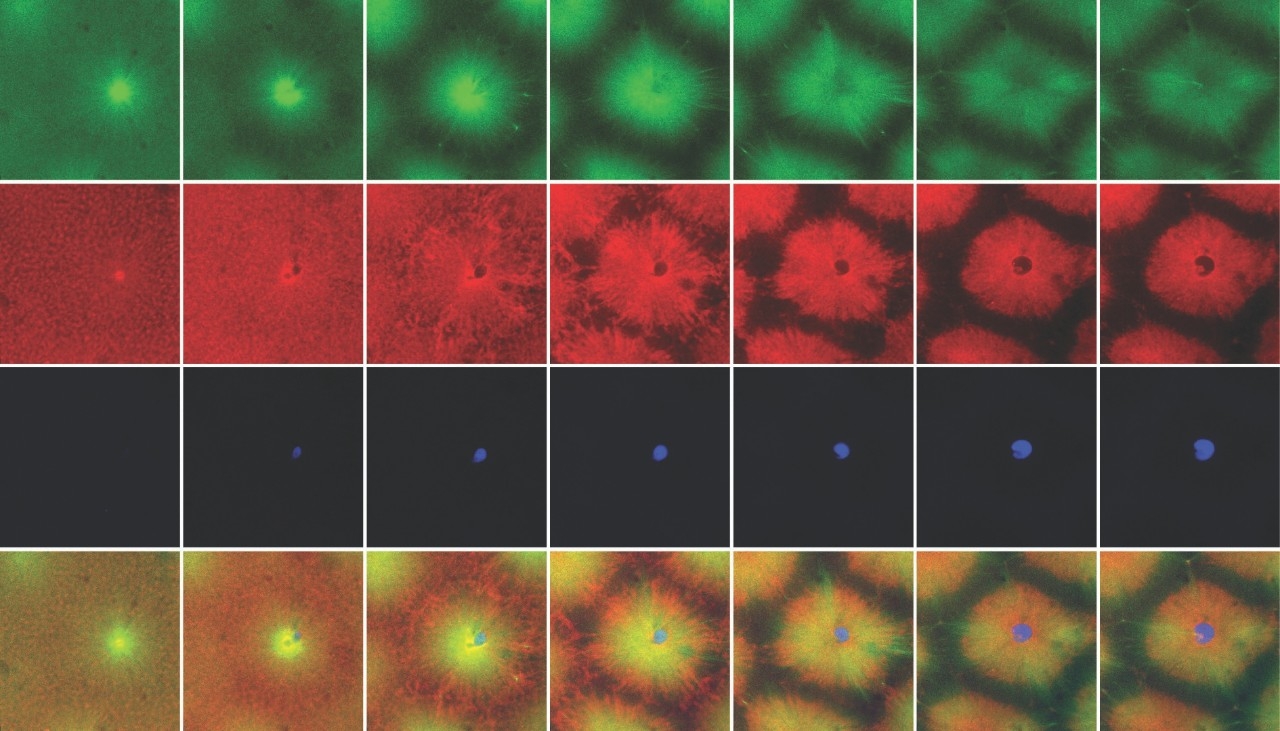
Smashing up frog eggs isn’t as permanent as it seems — the resulting goop can unscramble and form distinct cell-like compartments. “We were gobsmacked,” James Ferrell, MD, PhD, said of the discovery.
“If you blend a computer, you’d end up with tiny bits of computer, and they wouldn’t even be able to add two and two. But, lo and behold, the cytoplasm reorganizes,” said Ferrell, a professor of chemical and systems biology and of biochemistry.
Previous studies have shown that some of the tiny living machines inside cells can self-assemble from their purified components, but the new study provides the first example of self-organization at the scale and complexity of entire cells. And though these compartments fall short of real frog eggs — they can’t go on to form tadpoles — the discovery is eye-opening for scientists interested in learning how cells form or in creating synthetic cells.
Xianrui Cheng, PhD, a postdoctoral scholar, spotted the surprising behavior while performing experiments with frog egg cytoplasmic extracts consisting of the liquid contents of thousands of egg cells, as well as the nuclei — the organelles that normally serve as a cell’s command center.
“Initially, we were looking at a cell death signal spreading through extract in a long thin tube. And I noticed that a segment-like structure started to appear,” Cheng said.
After about 30 minutes, he saw that the nuclei were spaced evenly apart and boundaries had formed between them, resulting in the appearance of “sausage links” along the length of the tube.
Out of curiosity, Cheng examined samples of the cytoplasmic extract on cover slips through a microscope. He was shocked to see distinct compartments that resembled a sheet of cells.
“If you take the cytoplasm of the frog egg — note that the cytoplasm has been homogenized, so whatever spatial structure that was there has been completely disrupted — and just let it sit at room temperature, it will reorganize itself and form small cell-like units. That’s pretty amazing,” Cheng said.
To understand how this reorganization happens, Cheng added chemicals to disrupt the cytoskeleton, a network of protein filaments that extends throughout the cytoplasm. These tests revealed that microtubules, cytoskeletal filaments that provide structural support to cells, were required for compartments to form. Similar experiments disrupting motor proteins revealed that dynein, a protein that transports cargo along microtubules, was also required for proper microtubule localization.
“If you blend a computer, you’d end up with tiny bits of computer, and they wouldn’t even be able to add two and two. But, lo and behold, the cytoplasm reorganizes.”
James Ferrell, MD, PhD, professor of chemical and systems biology and of biochemistry
These cell-like compartments not only looked like cells; they divided like them too. The egg extract that the researchers used when they identified compartment formation contained a chemical that prevented the cells from entering the cell cycle. When this chemical was removed and sperm nuclei were added, the egg extract formed compartments that divided into smaller and smaller compartments.
Cheng and Ferrell shared these findings in an article published in the Nov. 1 issue of Science.
Their research suggests that the frog egg cytoplasm has the innate ability to generate the basic spatial organization of the cell and even has some of its functions. An open question, however, is what role this phenomenon plays in the normal physiology of the egg.
Such self-organization behaviors may be prevalent throughout biology. In the developing fruit fly embryo, thousands of nuclei initially share a common cytoplasm but ultimately form distinct cells and an organized body plan. Cells reorganize during mitosis, when structures break down and a single cell divides into two intact daughter cells.
As Cheng realized, there’s more to the cytoplasm than it seems at first glance.

