How aging might lower cancer risk
Young adult mice had more, larger lung tumors than elders
While our chances of getting cancer increase as we age, a recent Stanford Medicine-led study shows that changes related to aging might also suppress cancer.
In the study, researchers found that aged laboratory mice develop substantially fewer and less aggressive lung tumors than young adult animals.
“It’s a striking finding,” said Monte Winslow, PhD, an associate professor of genetics and of pathology and a co-senior author of the research with Dmitri Petrov, PhD, a professor of biology. “We would expect that older animals would get more and worse cancers, but that’s not at all what the study found. So, what is it about the molecular changes associated with aging that suppress cancer?”
To figure that out, the researchers examined the role aging plays in the biological mechanisms that transform healthy cells into proliferating cancer cells.
Studies in humans have shown that cancer incidence rises sharply beginning around age 50 and peaks around ages 70 to 80. This is because each time our cells divide, there’s a chance that mutations will be introduced into the DNA.
This leads to mistakes in how genes are expressed and function, and that can cause cancer. However, the curve plateaus or drops at age 85. While this could be due to decreased screening and diagnoses or because of a kind of natural selection — maybe people who live to a ripe old age tend to have immune systems better equipped to eliminate developing cancers — another possibility is that aging itself suppresses cancer development.
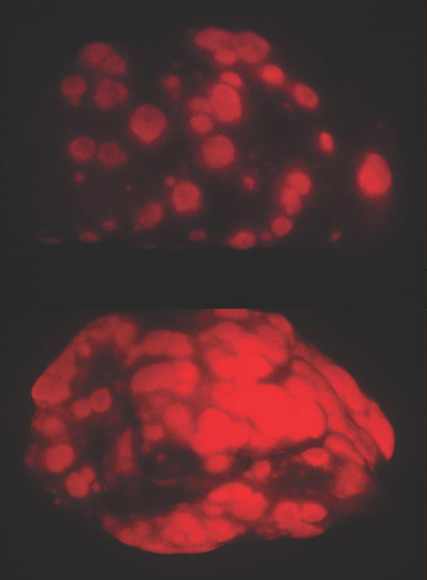
For the study, published in November 2025 in Nature Aging, former graduate student Emily Shuldiner, PhD, genetically engineered mice such that she could initiate fluorescently tagged lung cancers. Because she compared tumor formation in young adult (4 to 6 months) to old (20 to 21 months) mice, she had to wait nearly two years for the animals to age.
When they had, she induced lung cancer formation in the animals. Fifteen weeks later, the amount of cancer in the lungs of the younger mice — measured by lung weight and fluorescent imaging — was about three times as high as in the old mice. The younger mice also had about three times as many tumors than the old animals.
“In every way we could measure, the younger animals had worse cancers,” Shuldiner said.
To understand what was protecting the older animals, she investigated the effect of inactivating 25 tumor-suppressor genes in the lung tumors. These genes make proteins that normally block the development of cancers.
Although tumor growth increased when these tumors suppressors were inactivated regardless of age, the effect was greater in younger mice. Inactivating one tumor suppressor gene in particular, PTEN, had greater differential impact than the others.
“It suggests that the effect of any given mutation, or the efficacy of cancer therapies targeted at specific pathways, might be different in young versus old people,” Shuldiner said.
The findings illustrate the importance of developing new models of cancer that incorporate the effects of aging to develop new therapies.
“We develop animal models of cancer with an eye to developing new treatments for patients,” Winslow said. “But for this to work, the models have to be correct. And this study suggests that models using young animals might miss important aging-related changes.”

