Rocket men
Analyzing the breath of critically ill children at warp speed

In a dimly lit room, next to a supersonic jet engine test rig, three Stanford engineering graduate students sat around a whiskey bottle. All was quiet on this Friday evening in 2013 except for their lab’s visceral hum, a rumbling of fans, flames and gases rushing through jet-propulsion nozzles.
These three rocket-combustion experts — Christopher Strand, Victor Miller and Mitchell Spearrin — were talking about the future. In a few months they would have doctoral degrees, and then what?
As boys, all three grew up away from big city lights, with a clear view of the stars in the night sky. And they dreamed about rockets and exploring space.
Now that the space shuttle was grounded and its successor scrapped for being over budget, what would they do instead? Work at an aerospace company? Consult on military projects?
Extra
Watch Victor Miller and colleagues reveal the gas dynamics of lighting and extinguishing a match.
It was Strand who initiated a series of brainstorming sessions that challenged them to think beyond outer space.
“Somewhere between ideas on fixing San Francisco’s parking problem and inventing a marijuana Breathalyzer, we decided to see if we could use our education and expertise in combustion science to analyze human breath for disease,” says Miller.
After all, the human body is essentially a biochemical engine. It consumes fuel and exhales waste gases. Maybe the three of them could engineer a disease Breathalyzer? It would be a gadget straight out of Star Trek — a quick, noninvasive way to detect everything from diabetes to cancers.
Many have tried and failed to create such a device. But these guys are rocket men. They assume risks without fear. They achieve the impossible without breaking a sweat. They take giant leaps for mankind.
Sure, they didn’t know much about medicine, but they figured that with a little luck and a lot of hard work, they just might be able to do it. The first step was to find medical experts to help, so they contacted a group of pediatricians at Stanford’s medical school.
Saving Ethan
Five years ago, professor of pediatrics Gregory Enns, MD, was called into the neonatal intensive care unit at Lucile Packard Children’s Hospital Stanford to help a newborn in trouble.
The child’s mother, Tiffany Nguyen, was a business software consultant and his father, Luan Pham, was a systems engineer. They were immigrants from South Vietnam, excited about starting a family in the United States. After 18 hours of labor, their baby boy was born. They called him Ethan, a biblical name that means “enduring strength” in Hebrew.
But the morning after his birth, Ethan cried continually. By noon, his blood sugar and temperature dropped. His body became limp. The attending pediatrician couldn’t figure out what was wrong, so two days after Ethan’s birth, the infant was moved from a San Jose community hospital to Lucile Packard Children’s Hospital Stanford. That was when Enns, a biochemical geneticist who diagnoses and treats metabolic diseases, was contacted.
When Enns first examined Ethan, the prognosis was grim. Ethan’s tiny heart was beating erratically and his blood sugar level was dangerously low. Enns didn’t think the child would survive the night. But he put this possibility out of his mind and did his best. Enns, with his reassuring smile, quirky cartoon ties and clear blue eyes, is also a professional optimist.
First, the cardiac team was called in to help stabilize Ethan’s heart. Then Enns tried to figure out why Ethan’s blood sugar was so low. He suspected that Ethan had a genetic defect of the metabolic system. This could result in a buildup in the bloodstream of ammonia, a chemical that is normally detoxified by the liver. The blood test for this condition, called hyperammonemia, is slow and unreliable. Its analysis takes about an hour. By the time Ethan’s blood test came back, the level of toxic ammonia was almost 10 times higher than normal. Even if Ethan survived the next few days, he would always be at risk of another ammonia surge that could cause serious brain damage if not treated promptly.
Ammonia is a chemical byproduct released when the human body turns one type of fuel — specifically, digested protein molecules — into energy. The body eliminates this toxic waste by converting it in the liver to nontoxic urea, then sending it through the kidneys so it can be eliminated in urine. If anything goes wrong in this chain of organs and biochemical processes, ammonia builds up.
At this point there wasn’t time to do an in-depth genetic analysis to figure out what was wrong, so Enns expedited a biochemical blood test that revealed Ethan’s body was unable to digest long-chain fatty acids, a major component of breast milk and its precursor, colostrum. Because of this, Ethan’s body lacked enough energy to fuel his vital organs.
Maybe the three of them could engineer a disease Breathalyzer? It would be a gadget straight out of Star Trek — a quick, noninvasive way to detect everything from diabetes to cancers.
So Enns fed Ethan intravenously with a solution of high-calorie sugar and medium-chain fats. Then he administered a drug to remove the excess ammonia circulating in his bloodstream. Against the odds, this strategy saved Ethan’s life.
“Ethan was the sickest child in the intensive care unit I’ve ever seen turn around,” Enns says.
Once Ethan was out of danger, Enns sat down with the parents to talk about the realities of caring for a child with a metabolic disorder. It requires constant vigilance. They have to protect Ethan’s metabolism from stress, especially viruses. And they have to be alert to signs of lethargy and confusion — indications of high ammonia levels. If they suspect an excess of ammonia, a life-or-death drill will be initiated. Rush to the hospital.
Watch a phlebotomist poke the child with needles for the blood tests. Wait an hour for test results. If the result is high, hospital staff will administer ammonia-grabbing drugs and intravenous fluids, retest the blood and repeat as needed. Delayed treatment could lead to permanent brain damage or even death.
Enns has a superhuman ability to connect with patients and families in these difficult situations. Most of the children he works with have extremely rare diseases, for which research is limited and treatment plans are based on comfort care, guesswork or some combination. He is able to talk with a 10-year-old with severe developmental disabilities at exactly the right level, then turn to offer advice to parents on health insurance issues, never lapsing into technical doctor-speak.
When asked how he protects himself from the emotional stress associated with these conversations, Enns points to his prematurely silver hair and says, “I don’t.”
This was the dilemma for Enns: He could save these newborns, but then what?
Hope came out of the blue two years later, when he received a call from David Stevenson, MD, senior associate dean for maternal and child health at Stanford. Stevenson told him he knew of three Stanford rocket engineers with a novel idea for analyzing human breath, and they were looking for a medical condition to try it on. Would Enns collaborate with them? Enns immediately thought about patients like Ethan, and he jumped at the chance to help.
“Maybe these engineers could succeed where many others had failed,” says Enns. “I thought, after all, they’re rocket scientists.”

Mission control
The idea for the disease breath analyzer was born in Stanford’s High Temperature Gas Dynamics Laboratory. This lab, tucked into an unobtrusive, sandstone-and-tile building behind Stanford’s Main Quad, has served as the launch pad for almost 100 combustion engineers, all of whom earned their doctorates under the mentorship of mechanical engineering professor Ronald Hanson, PhD.
In 2013, two of Hanson’s students — Christopher Strand and Victor Miller — sat at adjacent desks overlooking a “Rockets of the World” poster. They were both finishing dissertations on supersonic combustion ramjets, called scramjets for short. Strand was working on better ways to measure engine gas mixtures using lasers. Miller was developing gas-flow visualization techniques using lasers and high-speed cameras.
Scramjet technology, conceptualized in the 1950s, still presents researchers with extreme technical challenges. These engines use atmospheric oxygen to burn their fuel rather than having to carry liquid oxygen along for the ride. This allows scramjet-equipped craft to fly at speeds of more than five times the speed of sound. Theoretically, aircraft equipped with these engines could fly anywhere on Earth within 120 minutes. Scramjet space planes could carry greater payloads and operate more efficiently.
Strand, now 30, tall and lean with British-schoolboy wavy brown hair, has always wanted to be an astronaut. He was raised on a small farm in rural Alberta, Canada, the son of a single mother who worked as a bookkeeper. Strand didn’t apply to college during high school. But when he accompanied his girlfriend (now wife) to her first day of classes at the University of Alberta, he realized he’d made a horrible mistake.
“All of a sudden I knew that I belonged at a university,” Strand says. So that week, through a fortuitous connection, he met with the dean of engineering and talked his way into the school’s engineering-physics program.
Miller, 28, with mischievous eyes and the energy level of someone who just downed a triple espresso, is a fix-anything guy with a penchant for testing boundaries. He’s also a drummer in a ’90s cover band called Cloning Dolly. He grew up in Watertown, Wisconsin, a small town an hour east of Madison. His father was an ex-Marine-turned-engineer and his mother was a travel agent. As a boy, he was obsessed with airplanes. He graduated from Cornell University, summa cum laude, in mechanical and aerospace engineering.
Miller’s worldview had been influenced by a year in Stanford’s Accel Innovation Scholars program, which gives 12 PhD students access to entrepreneurial leaders in Silicon Valley. This program encourages bright engineering scholars to explore ways to apply their knowledge to some of society’s biggest challenges. In other words, Miller had absorbed the culture of Stanford entrepreneurship.
Strand also felt the allure of inventing a Silicon Valley “new new thing,” he says. “I think Vic and I empowered each other to pursue breath sensing. There is a certain confidence that comes with having a partner.”
When the team first began looking into the breath analyzer idea, a search of scientific literature revealed that breath testing with the human nose has been used in medicine since ancient times. The rotten-apple smell of acetone is a sign of diabetes. The smell of putrid socks is associated with kidney problems. A fishy smell is indicative of liver disease. Though these nose-based diagnostic skills are still used by some clinicians today, many researchers have recognized the opportunity to develop a medical device that could transform this art into a science.
The late Nobel laureate and Stanford chemistry professor Linus Pauling was one of the pioneers of modern breath testing. In the 1970s, he used a gas chromatograph to detect several hundred volatile organic compounds in breath, providing the first evidence that it is a more complex mixture of gases than anyone had imagined. Since then, more than 3,000 compounds have been detected. And though the signatures of ingested chemicals, like alcohol, may be easy to measure, it’s much more difficult to detect disease biomarkers, unique combinations of small molecules that may be present only in trace quantities in human breath.
The engineers figured that the technology they used in rocket testing, laser absorption spectroscopy, would be sensitive enough to make measurements of trace compounds in the breath.
For detecting gases in combustion flows, the technology works like this: A laser beam at a specific frequency is fired across a stream of burning gases, and a sensor on the other side of the beam measures the quantity of light that is transmitted through the gases. From this information, gas properties like temperature, velocity and the chemical composition of the exhaust gas mixture can be identified almost instantaneously. Just as engineers can use these data to tell if an engine is operating efficiently, they could tell if a human “engine” is operating in a healthy range.
To analyze the gases in human breath, Miller and Strand realized they’d need a laser that emits light in the mid-infrared frequency range. They also needed someone experienced in this range, and luckily, there was just such an expert on the other side of the rocket lab: Mitchell Spearrin.
All systems go
Spearrin’s life trajectory was set when he watched a rocket from Cape Canaveral soar over his home near Bryceville, Florida, population 3,000.
“I wanted to be an astronaut,” says Spearrin, 31, who is married with two daughters and looks like someone who might be cast as a square-jawed hero in a Hollywood blockbuster.
Although the U.S. space program was nearby, this career goal seemed light years away from his small, rural town. As a kid, he focused on sports and became the captain of his high school football and baseball teams, a natural leader. He also was good at math and graduated as the straight-A valedictorian of his senior class.
Encouraged by his parents, an elevator mechanic and a stay-at-home mom, Spearrin was determined to be the first in his family to attend college. He also hoped to play sports at the collegiate level, and it was through football that he found himself unexpectedly recruited by Harvard late in his senior year. In a matter of weeks he went from never having considered an Ivy League school to committing to Harvard’s football program and, in turn, an education he could not have fathomed. It eventually led him to Stanford’s mechanical engineering doctoral program.
During his time at Harvard, Stanford and a stint at aerospace manufacturer Pratt & Whitney, Spearrin fell in love with rocketry. “These machines represent a certain pinnacle of engineering: rockets control a convolution of physical extremes with a precision driven by intolerance for human error,” he says.
Spearrin, who at one point was voted by his Harvard football teammates as “most likely to start a business,” liked the idea of the breath analyzer, so he joined the effort. And at that point, they had a team in place. Strand knew about lasers. Miller knew about gas handling and photonics hardware. Spearrin knew about rapid analysis of gases using mid-infrared lasers. And Enns agreed to be their medical research mentor.
They started off the project with two roundtable discussions that included Enns, Stevenson and several other pediatricians. (Stevenson had worked on breath analysis of bilirubin, a chemical that can signal jaundice in newborns, early in his career.) They discussed the most urgent clinical needs for newborns, and ammonia screening rose to the top. A second priority would be to detect acetone, a diabetes marker, in newborns.
The graduate students then wrote a three-page proposal for their breath ammonia analyzer and submitted it for a pilot grant from Spectrum, a Stanford program that funds researchers with bold ideas for addressing important health-care problems. (Primary funding for these grants comes from the Spectrum Clinical and Translational Science Award from the National Institutes of Health.) They were awarded $49,000 to launch the project and teamed up with an industry mentor, Darlene Solomon, PhD, senior vice president and chief technology officer of Agilent Technologies. Then the countdown began. They had a year to get a prototype working.
“I thought it was a simple, elegant solution — though at the time, it seemed as if was too simple to actually work, given the small quantities of ammonia they were trying to measure within the complexity of human breath” says Solomon.
The engineers figured that the technology they used in rocket testing would be sensitive enough to make measurements of trace compounds in the breath.
The project got off to a slow start. The funds were delayed, and all three engineers had rocket science “day jobs” to work around.
They began with a schematic on how their device works. A person blows into a tube and breath gases are collected in a pressure-regulated cylinder that directs a controlled gas stream across a mid-infrared laser beam. When the beam hits ammonia, the molecules absorb specific wavelengths of light. A photodetector measures the amount of light that passes through the ammonia, then custom software calculates quantities of ammonia and plots it on an easy-to-read graph on a laptop computer. The device also measures carbon dioxide as a way of telling the software that one breath cycle is complete and another one is beginning.
The first prototype used a clear quartz tube for the gas cylinder, which Miller purchased for $50 on eBay from an equipment salvager in Austin, Texas. The breathing tube was attached to one end of the cylinder. Flow meters, pumps and valves were attached to the other end, all scavenged from the rocket lab. These would direct the gas stream across the laser beam.
Optical mirrors directed the laser beam onto the photodetector. Initially, the prototype was built on an 8- by 4-foot table with a Rube Goldberg array of gas-handling tubes, pumps and pressure gauges sprawled above and below. The team began to make its first measurements of breath ammonia, and during the first trial runs realized why no one had ever successfully developed an ammonia breath analyzer.
“Ammonia is a nightmare to work with,” says Spearrin.
Because the molecules are highly soluble in water and have an unstable electrical charge, they tend to stick to everything, including the inside of the human mouth and the walls of plastic tubing. So they switched to nonstick Teflon tubing. Temperature fluctuations distorted ammonia measurements, so an on-board heater and insulation had to be added to the device.
Finally, after six months of tweaking, the team brought its second-generation prototype into a quarterly grant-review meeting. The prototype was packed inside a custom box, which was placed on a wheeled cart. Beneath were a data acquisition system and various measurement instruments, all of which would be miniaturized into a more compact format in a commercial product.
A volunteer from the meeting blew into the tube, and a graph of the levels of ammonia and carbon dioxide in that given breath appeared on the computer screen.
Enns’ first impression of the rapid, easy-to-use device was “jaw-dropping amazement.”
We have liftoff
With the help of Enns, the engineers received Institutional Review Board permission to test their ammonia breath analyzer on human subjects, specifically two 16-year-old boys admitted to the hospital for hyperammonemia. These teens were representative of their target patient population — they were cognitively and physically impaired from ammonia surges. One used a wheelchair. Both spoke slowly, in broken sentences.
It brought home the importance of why the team was working on the breath analyzer project.
Their plan was to have the teens blow into the device’s breathing tube after each of their blood draws over the two or three days it would take to normalize their ammonia levels. But they soon realized that it was difficult to explain to the teens how hard to blow.
Finally, Strand figured out a strategy that worked. He gave the boy the tube and said, “Pretend that this is your elephant nose and make a sound like an elephant.”
This insight prompted the team to redesign the software to provide visual feedback that showed patients when they were blowing hard enough. They also started designing a passive, under-nose breathing tube that could be used without active blowing, which will be necessary for some patients but requires more sensitive detection.
Patient testing also refined their thinking on the technological advantage their device brings to the field. The major weakness of the ammonia blood test is that by the time the results are received by a treating physician, it is hour-old information that may not represent the true ammonia levels of a patient. The breath analyzer enables super-fast, repeatable testing so ammonia levels can be verified and treatment can begin immediately.
“Babies breathe so fast that it’s hard to get an accurate ammonia reading using a device with a slow response time,” says Spearrin. “What our device is really good at is rapidly measuring intra-breath dynamics, showing how the chemical composition of a breath changes over time.”
In just a year, the team had gone from a rough idea on paper to a working prototype, patient-tested. This is warp speed in the medical device world. They are also preparing articles for publication describing the underlying spectroscopy, the device and, ultimately, their clinical studies.
Spearrin didn’t realize how hard this project was supposed to be until he called a respected expert on hyperammonemia for advice. Before Spearrin could ask his questions, the expert said, “You’ve chosen a horribly challenging project because ammonia is the most difficult molecule to measure and newborns are the most difficult patient population to work with.”
Spearrin replied, “But we’ve already built a working prototype and we’ve tested it on two patients.”
In the fall of 2015, the team is planning a second, larger patient trial that will involve younger children. There’s a good chance Ethan will be in that trial. Since they finished their first prototype, they’ve received grants from the NIH’s Small Business Technology Transfer program and the Wallace H. Coulter Foundation. The Stanford Office of Technology and Licensing has filed a provisional patent, and the team has formed a company, Lumina Labs. The company, funded by the NIH small business grant, has established a research consortium with Enns and Stanford.
“What impressed me about this development team is that they really listened to all the advisers’ technical concerns, methodically addressing each one. And they did so while still getting a prototype into testing amazingly quickly,” says Solomon.
Waiting to exhale
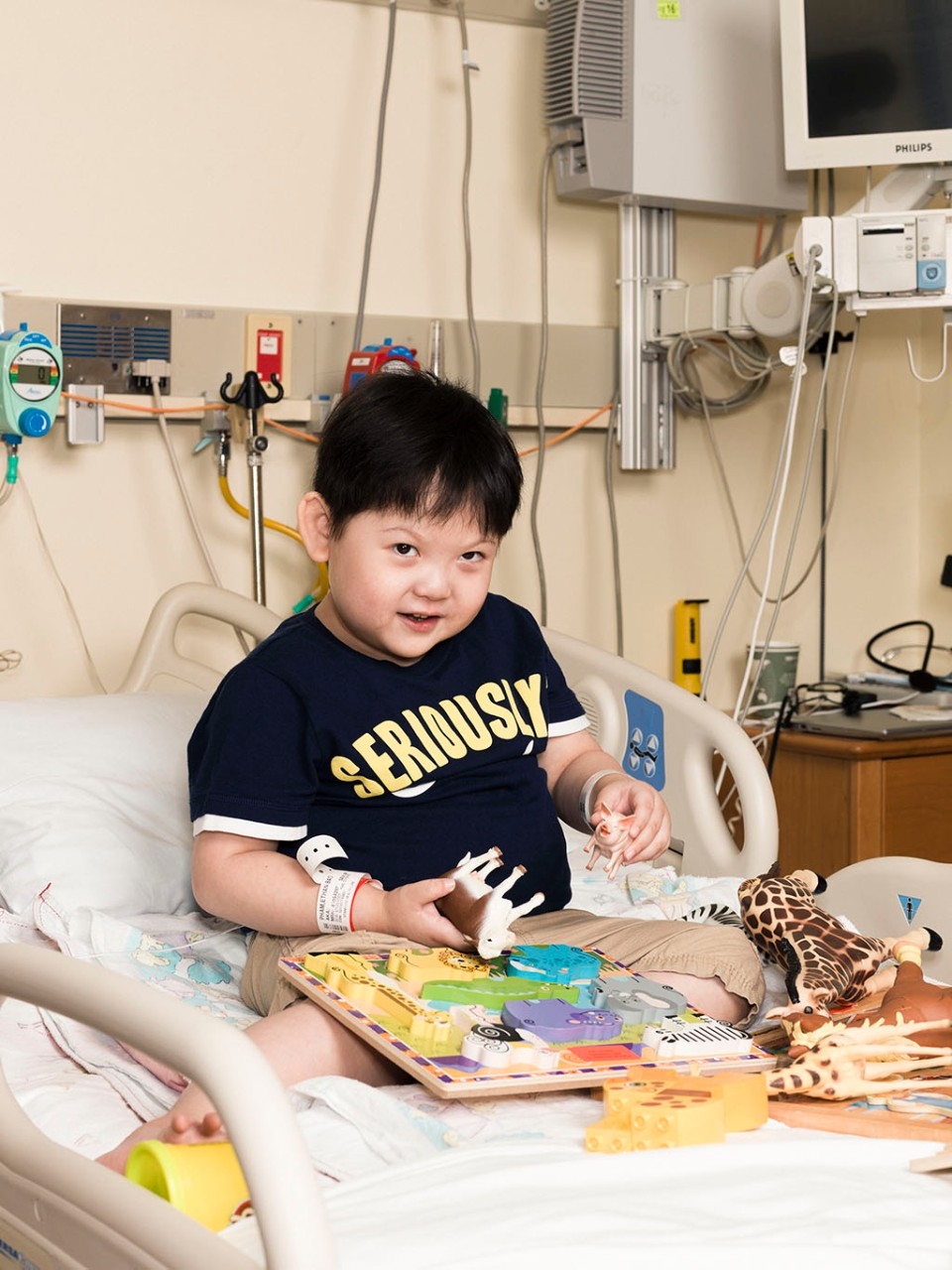
Five years after his birth, Ethan Pham, with chubby cheeks and bear-cub ears, looks and acts like a typical kindergartener. His mother — a halo of dark hair framing her ivory face — plays with him as he sits in his hospital bed, happily singing with cartoon farm animals on TV. On the bed tray is a sheet of paper where he has practiced writing his name with crayons.
Ethan is recovering from a surgical procedure to insert a tube through his chest into an artery of his heart. This permanent IV port will make it easier for the care team to quickly administer ammonia-grabbing drugs when needed. In the past, a nurse would have done this by inserting a syringe into an arm blood vessel, but with so many pokes over the years, it became hard to find an undamaged, free-flowing vein. He’s also under observation for high, unexplained fluctuations of ammonia.
It takes a dedicated team to keep Ethan alive. His family, schoolteachers and medical practitioners are continually on the lookout for signs of high ammonia levels. Episodes can happen at any time. Each incident means a 30-minute drive to the critical care unit, where staff members stand ready to draw blood. Ethan’s medical team — his pediatrician, Rebecca Fazilat, MD, at Sutter Health San Jose; Enns; and the hospital staff at Stanford Children’s Health — is on call 24/7.
Many times the ammonia blood tests, which can be done only at the hospital, are wrong or ambiguous. If the test is positive, it typically takes a day or two in the hospital to normalize the ammonia levels, with repeated blood tests every few hours. Sometimes the family is halfway home when a nurse calls them back to redo a test. Ethan has spent about half of his kindergarten year in the hospital.
Ethan’s teachers have been trained to accommodate his condition. His work areas must be extra clean and sick kids need to be kept away. His diet is carefully monitored — no birthday cake, since he can’t digest it. Ethan doesn’t have the muscle strength to climb on the playground equipment, so he often sits on the side, playing with his plastic farm animals or trying to kiss Catherine, a girl in his class he really likes.
Nguyen and Pham, like most parents who have children with metabolic defects, are perpetually fatigued. When Ethan is in the hospital, Nguyen stays by his side and her husband joins her after work. They often eat dinner at the hospital cafeteria. Nguyen’s parents and sister live close by, and they help out when they can. For Nguyen, it’s a full-time job keeping Ethan from slipping into an ammonia-induced coma.
What keeps them going is their faith (Nguyen is a Catholic and Pham is a Buddhist) and the hope that someone, maybe even the rocket men, will find a better way to test ammonia levels in children with metabolic diseases at the hospital and at home. This would allow Ethan, with his enduring strength, and his family to live a more normal life.
Blue-sky thinking
It’s worth looking at the breath analyzer project and asking, what can fuel more of these big ideas in medicine?
Spearrin recently summed up what motivated his team: “For us, it’s not that ammonia sensing is the perfect challenge. It’s that the breath analysis field is underdeveloped. We’re leaders in this particular gas-analysis technology, and there are clinical researchers here at Stanford really open to collaborating with us. It gives us a chance to make a significant contribution through cross-disciplinary efforts.”
What worked was to empower an ambitious team of young engineers to look at an old medical problem with fresh eyes. They were given starter funds to try out their big ideas without fear of failure. There was institutional buy-in, making it acceptable for people outside of the medical system to observe, ask questions and change the way things have been done in the past. And they were given access to mentors who could inspire them, help remove bureaucratic roadblocks and keep them from making big mistakes.
Strand adds, “Being in a clinic and working with kids gave me a unique sense of purpose that I haven’t felt in my research before. I’ve had the good fortune of getting to be part of a lot of exciting and challenging research, but never where the need is so tangible, urgent and, most certainly, so personal. It makes a difference if this problem is solved today instead of tomorrow.”
Of course, anyone familiar with medical device development would be quick to add that there’s a tremendous amount of work to be done before the ammonia breath analyzer is widely available. There need to be more prototypes. Clinical trials. Independent validations. But one thing we all can probably agree on is this: Medicine needs more rocket scientists.

