Pathways
From the eye to the brain
Carla Shatz , PhD, remembers the reviews well, or rather the lack of them. It was 1997, and she and her postdocs had just discovered that a molecule ordinarily associated with the immune system played a role in shaping young, developing brains.
Related reading
Studying how we see is changing views of decision-making, autism and neurological disease
It was a big discovery, so they tried to publish it in a major scientific journal. But the editor — after consulting briefly with a couple immunologists, who told him Shatz and her team must have made a mistake — rejected it.
Her postdocs got discouraged, but Shatz had a different reaction. “I got angry.” Shatz and her team had made a breakthrough discovery, and she was going to make sure the work got attention.
She hadn’t set out to stir up trouble. Four decades ago, Shatz just wanted to know more about how the visual part of the brain wires up during development. But as she pursued that goal, she and her team made some of the past 40 years’ most important — and at times controversial — discoveries about brain wiring during developmentally critical periods. Driven by curiosity and a refusal to be bound by traditional thinking, Shatz has time and again found herself in uncharted and fertile territory.
Seeing inside the brain
Shatz’s first major discovery emerged in the 1980s, not long after she joined Stanford’s neurobiology faculty, and built on her earliest work as a student and postdoctoral researcher on the development of the visual system. By that time, she was already a trailblazer in one respect: In 1976, she had become the first woman to receive a PhD in neurobiology from Harvard University, and in 1978, she became one of the first women to join the basic science faculty at Stanford’s School of Medicine.
In mammals with good binocular vision such as cats, the visual system is a precision instrument, organized so that light picked up at neighboring points on the retina at the back of the eye is projected onto neighboring neurons in the brain’s visual cortex. The brain also does an extraordinary job of separating out information from the left and right eyes, and it does these jobs so well that some assumed the organization was hardwired and took shape independent of sensory input. The finest details, these researchers thought, were explicitly preprogrammed in an animal’s — or human’s — DNA.
Shatz suspected otherwise. When the brain first forms in the womb, it is not the finely tuned machine it eventually becomes. Instead, its connections are a poor approximation of what they will grow into, and, sort of like a haphazardly wired computer, it does not operate at its full potential. But as brains develop, something happens to prune and fine-tune the wiring into a precise computer that can translate light shining on the eyes into an understanding of the world around it.
Shatz’s investigations into pruning and tuning led to a 1988 study in which she and Michael Stryker, PhD, injected a drug, tetrodotoxin, into the developing brain. Tetrodotoxin blocks electrical activity in neurons, and in this case, it also blocked normal pruning in the visual system.
“The neurons are spontaneously active, but it’s not like twinkle twinkle little star, and it’s not like a big tsunami either.”
To Shatz and Stryker, the message was clear: Although the rough structure of the visual system is programmed in our genes, the precise connections among neurons in the adult visual system are not explicitly predetermined. Instead, those connections emerge out of an interaction between genes and neural activity. Specifically, Shatz says, neural activity regulates the genes that are ultimately responsible for pruning and strengthening connections between neurons.
Those results eventually led Shatz to coin what may be the best-known aphorism in neuroscience, “neurons that fire together wire together,” and its corollary, “out of sync, lose your link.” In other words, where there is electrical activity in the brain, connections between neurons are built up, while others are pruned away.
Although that idea is now universally accepted, that acceptance did not come easily. Mostly in conversations but also in a few published studies, Shatz says, neuroscientists argued that the results were a mistake. Shatz and Stryker, these researchers argued, had probably just poisoned the brain. Tetrodotoxin is, after all, a toxin.
There were other questions as well. To produce organized visual circuits, electrical signals had to be coordinated — according to the “fire together, wire together” aphorism, organized connections require organized signals — but no one knew what was responsible for the organization. Shatz and her team still believed that coordinated electrical activity was crucial to the proper development of vision circuits, but they would need to do more to prove it.
Waves on a sea of neurons
Seeing begins when light shines on our eyes, triggering neurons in the retina to fire, which sends electrical signals into the brain’s vision centers.
The funny thing is, animals in the womb like those in Shatz and Stryker’s experiment, cannot see. They are still in the dark, and their eyes are closed. In fact, the rods and the cones in the retinas are not yet fully formed, meaning that even if light did somehow reach their eyes, it could not trigger any neural signals via electrical activity. So if vision circuits need coordinated electrical signals to develop properly, where do those signals come from, and what coordinates them?
The answer came in the early 1990s when Shatz and her postdoctoral researcher Rachel Wong, PhD, linked up with Denis Baylor, MD, then a professor of neurobiology, and his postdoctoral researcher Markus Meister, PhD. Wong was a physicist and an expert on the retina, and Baylor and Meister had been developing electrode arrays that could measure activity in around 100 neurons at a time. That made it possible to study what was going on in developing retinas.
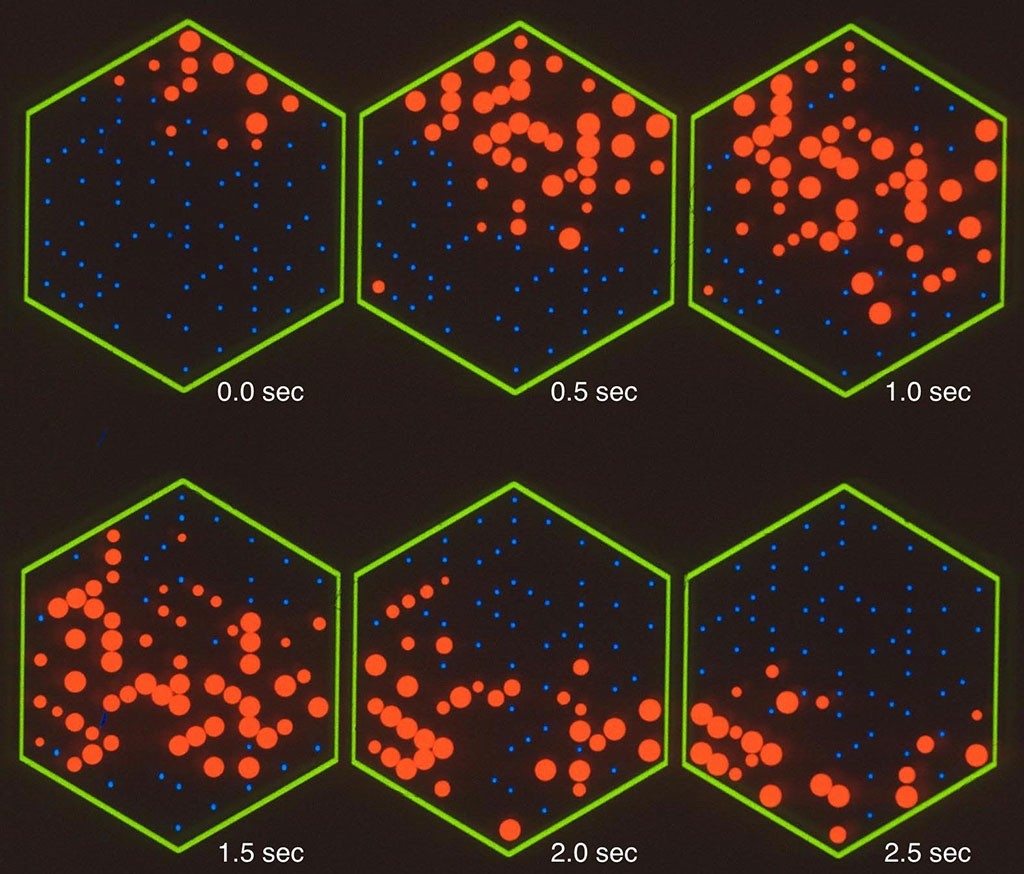
Even in newborns without fully developed retinas, the foursome reported in a 1992 paper, intricate networks of neuron circuits had already formed across their retinas.
Those neuron networks turn out to be critical. Neurons can fire at any time, more or less at random, but without connections to other neurons, nothing interesting happens. With connections, neurons can send signals to other neurons across the retina. And with the networks that Shatz and colleagues found, something beautiful happens: One neuron fires, triggering nearby neurons to fire, forming a kind of pulse of electrical activity — and then, the pulse starts to travel around the retina in wave-like patterns before fading away. Then another neuron fires at random, and the waves begin again.
“The neurons are spontaneously active, but it’s not like twinkle twinkle little star, and it’s not like a big tsunami either. It’s these local waves,” Shatz says — exactly the kind of coordinated activity a developing visual circuit needs.
Shatz moved her lab to the University of California-Berkeley in 1992, but kept up the work on what she called retinal waves. Later studies using a technique called calcium imaging revealed those waves in captivating detail, yet there were still critics. One anonymous reviewer suggested they were just witnessing “spreading depression,” a wave of out-of-control, exploding neurons — which made little sense, Shatz says, since spreading depression destroys neurons, while retinal waves bounce around, fade away, and reemerge over days and weeks.
Other critics noted that the experiments were not done in living animals — the electrode arrays were too big and heavy to make that practical. Instead, Wong, whom Shatz describes as preternaturally handy, gently removed the retinas and studied them in a dish. In 2014, researchers finally succeeded in recording retinal waves in live animals — 20 years after Wong, Meister and Shatz published their first results.
What’s that molecule doing there?
In the meantime, Shatz started to wonder how exactly the brain implements its “fire together, wire together” and “out of sync, lose your link” rules, so she and her lab decided to screen mice for genes that might be involved in strengthening and pruning links between neurons. After all, she reasoned that even the “fire, wire” rule must have a genetic basis.
What they found was astonishing: Neurons’ electrical activity governed the production of a group of molecules, called major histocompatibility complex class one. MHC1 molecules are best known for their role in the immune system, and according to widely held views at the time, MHC1 was not even supposed to be in the brain under normal circumstances, let alone involved in shaping the structure of the visual system.
When the paper was rejected, Shatz felt bad for her postdocs, Roderick Corriveau, PhD, and Gene Huh, PhD. “I mean, they had done really beautiful work,” she says.
The team persisted, and less than a year later — thanks in part to a few more experiments and an intrigued editor, Shatz says — they published their study in Neuron in 1998. In 2006, her team — now at Harvard — identified a molecule, paired-immunoglobulin-like receptor B, or PirB, that resides on neuron surfaces and works with MHC1 molecules to drive synaptic pruning like lock (PirB) and key (MHCI). When MHC1 lands on PirB, it acts like a switch that prevents new synapses from forming and triggers the pruning of others.
“It did seem a little surprising,” says David Raulet, PhD, an immunologist at UC-Berkeley who provided the MHC1-suppressed mice for Shatz’s lab when they were based at Berkeley. Back then, he says, “we just didn’t know where to put it” in the context of the immunological canon.
The role MHC1 plays in healthy brains remains bemusing, if not exactly controversial, in immunological circles — but no matter, Shatz says. By then, “we were off.”
Disease and the pursuit of basic — and interdisciplinary — science
Shatz had moved to Harvard University in 2000 to chair its Department of Neurobiology, where she continued to learn more about how MHC1 and PirB worked. Armed with that knowledge, she turned toward something that seemed remote at best when she first started out: disease. In 2004, Shatz and postdoctoral researcher Lisa Boulanger, PhD, proposed that MHC1 could play a role in neurodegenerative diseases such as Alzheimer’s, in which the brain’s ordinary pruning mechanisms go haywire.
In 2007, Shatz moved back to Stanford and, since then, her lab has shown that suppressing PirB protects against memory loss in mice that have been genetically modified to carry the human Alzheimer’s gene. In 2012, they showed that suppressing MHC1 could help protect the brain from the worst effects of a stroke, a finding that may inspire new treatments for a variety of neurological disorders, or at least a better understanding of them.
“Carla literally changed the way we think about development of the visual system,” says William Newsome, PhD, professor of neurobiology and director of the Stanford Neurosciences Institute. “But beyond that, it turns out that many of the mechanisms Carla discovered hold true for the development of other pathways in the brain, not just the visual pathways.”
Today, neuroscientists have come to accept most of her conclusions. Shatz, a member of the neurosciences institute, is now the Sapp Family Provostial Professor and directs Stanford Bio-X, a hub of interdisciplinary collaboration on campus. She was elected to the National Academy of Sciences in 1995. In 2016, she won the Kavli Prize in Neuroscience, among the field’s highest honors.
Still, Shatz says, she worries for the future. Throughout her career, she says, she has benefited from interdisciplinary work. A major factor in her return to Stanford was the chance to lead Bio-X, where she could help foster — and, more to the point, fund — occasionally bonkers-sounding collaborations between doctors, engineers and basic scientists that might not happen any other way.
And, she says, her career benefited from the ability to focus on basic science without regard to the drugs or therapies that might result. She laments the pressure on a younger generation of researchers to translate their research into treatments as quickly as possible.
“It’s crazy to think that just by studying fundamental aspects of the development of the visual system, it’s led us to think in new ways, potentially really important clinical ways, about not just treating visual problems but possibly problems of synapse loss as occurs in Alzheimer’s or Parkinson’s or even multiple sclerosis. You never know where basic research questions will lead.”

