Brain balls
Tiny lab-grown blobs could aid understanding of psychiatric and neurological disease
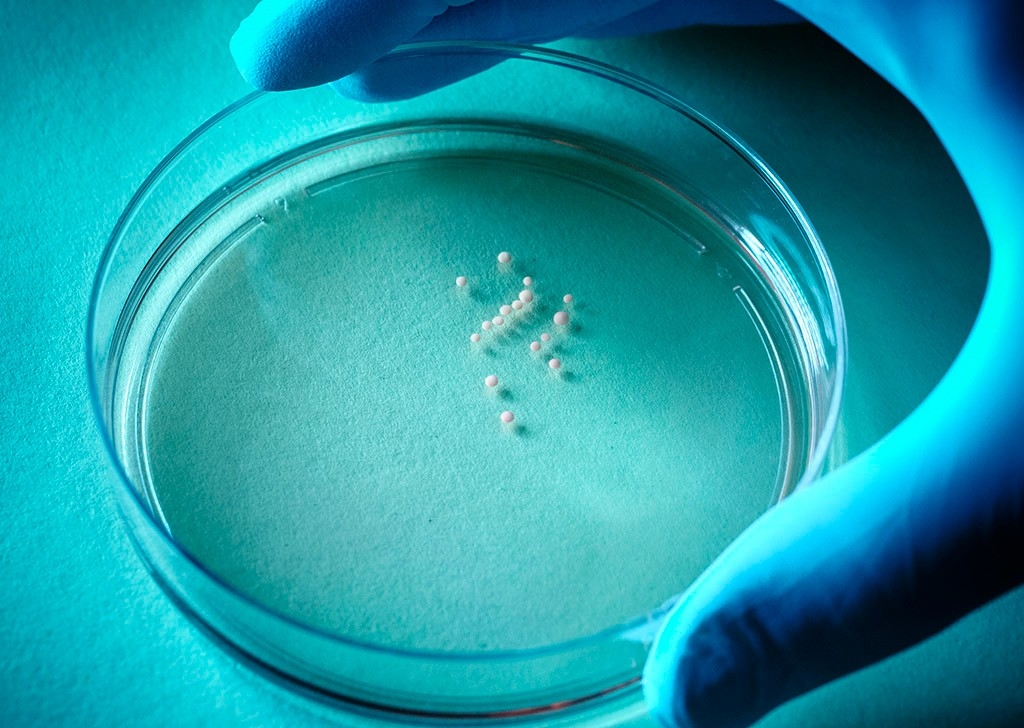
A brain in a bottle, hmmm? And it’s helping a Stanford neuroscientist do his research?
C’mon. You’re kidding, right?
Exaggerating, maybe. Kidding, no. And we’re not talking about computers. A Stanford neuroscientist is growing brainlike blobs in dishes, and they’re helping him learn a whole lot about his favorite subject.
A brain is a complicated thing — the most complicated thing in the universe, some say — with close to 90 billion nerve cells, or neurons, and some 150 trillion individual neuron-to-neuron connections, called synapses. So, not such an easy entity for researchers to wrap their heads around.
Get out your 3-D glasses, because what follows reads like science fiction: Suppose you hope to learn what goes wrong during early brain development. One could learn a great deal about an individual’s neurodevelopmental condition by studying that person’s neurons close up, at the molecular, cellular and circuit levels. But how? You can’t exactly scoop a chunk out of someone’s living brain. And dead ones don’t tell you nearly enough.
Is there a workaround? A team led by Sergiu Pasca, MD, assistant professor of psychiatry and behavioral sciences, has found one — a technique that reliably and selectively produces pinhead-sized replicas of specific, different human brain parts in laboratory dishware. While researchers had previously developed other ways to culture cells to form brain organoids — minute clumps of tissue enriched for brain cells — these organoids also contained stray cells from other parts of the body, and they lacked the structural organization that characterizes components of a real brain.
Pasca’s method is notable because the clumps he’s growing contain only cells that are supposed to be in the brain, and because, rather amazingly, the clumps’ structures actually recapitulate those of distinct brain regions. His technical term for these little brain balls is brain-region-specific spheroids.
Pasca published his method for creating the brain balls in 2015 and knows of a dozen laboratories around the world that have successfully created them since then. Based on the hundreds of questions he has received about the protocol, he assumes there are many more.
The spheroids enable researchers to zero in on the pathological mechanisms that disrupt fetal brain development in autism, epilepsy and other neurodevelopmental disorders. They can also help neuroscientists understand the causes of faulty brain development in prematurely delivered babies.
“This is our doorway into personalized psychiatry,” says Pasca.

A brain ball is not a brain. The constructs are, at present, devoid of some important cell types found in a real human brain. They receive no sensory inputs from the outside world, and they can’t initiate muscular contractions. They also lack blood vessels, whose absence means a brain ball has to get its nutrients only at its surface, limiting its size.
Scientists are far from being able to grow a brain in a bottle. But as researchers learn to create more complex brain organoids and consider transplanting them into animals, ethical quandaries will multiply.
Pasca’s interest in research all started with chemistry experiments in his childhood home in Transylvania, a fabled region of Romania where he was born in 1982. He set up a chemistry lab in his parents’ basement at age 11 and promptly presented them with his first product: a crater in their backyard. His mastery of the subject improved, and in his final year in high school he won a national chemistry competition and a free ride to a nearby medical school. There, he met the woman who is now his wife, Anca, in a microbiology class. She was from Transylvania, too.
“I was born 30 miles from Dracula’s castle,” says Anca Pasca, MD, a clinical fellow in neonatology in Stanford’s Department of Pediatrics. Sergiu Pasca realized early in his medical training that his primary interest was research. But the school he was attending was so resource-poor that his biochemistry professor had to tap her own salary to fund his project: analyzing numerous substances in the blood of children with autism in search of a biochemical signature.
“I needed blood,” recalls Pasca, “so I would approach parents outside of a treatment center and ask them if I could get samples from their kids. You might expect the parents to be a little suspicious — after all, this was Transylvania — but instead these people would cry and hug me and thank me for working on this disease.”
Studying autism up close
Eventually, Pasca decided that “blood is pretty far from the brain.” He wanted to study autism up close, at the level of the neuron. And he knew exactly where, and with whom, he wanted to study it. He came to Stanford in 2009 to pursue a postdoctoral fellowship in the lab of Ricardo Dolmetsch, PhD, then an assistant professor of neurobiology and now global head of neuroscience at Novartis Institutes for Biomedical Research in Cambridge, Massachusetts. Dolmetsch had redirected his research to autism spectrum disorder after his son was diagnosed with it.
“Coming from an unknown medical school in Romania, without any molecular biology experience, and barely able to speak English, I thought I had no shot at joining such a successful lab,” Pasca says. “But Ricardo was looking for somebody with an interest in autism, and he gave me a chance.”
When Pasca started his postdoc in 2009, researchers were exploring ways to grow specific cell types from induced pluripotent stem cells, which had recently been discovered. Like embryonic stem cells, induced pluripotent stem cells (known as iPS cells) are capable of differentiating into virtually all the body’s different cell types.
But unlike embryonic stem cells, iPS cells can be obtained, with relatively routine laboratory procedures, from any person’s skin. Dolmetsch wanted to generate neurons derived from the skin of a patient with a rare form of autism called Timothy syndrome that is caused by a genetic mutation. Pasca signed on.
In 2011, Dolmetsch and Pasca succeeded, which allowed them to pinpoint a mutation-induced physiological malfunction responsible for the symptoms of the disorder. Theirs was the first model of autism built from neurons that mirrored those in the brains of patients. The scientists hoped to monitor these neurons’ development over a longer term.
“But the two-dimensional cultures we were using were too constrained,” says Pasca. “The cells didn’t act quite as they would in a human brain. Most 2-D cultures disintegrate after 100 days or so. But neurogenesis in the human cortex is complete only by 26 weeks of gestation, and astrocytes, brain cells that are absolutely essential for making working synapses between neurons, don’t even start getting made until late in gestation.”
‘We started wondering, what if we let these neural progenitor cells just float, suspended, in the dish instead of letting them attach?’
Pasca resolved to improve the culture situation. That summer, he began noodling around with what he calls a “Saturday experiment” of trying to perfect a three-dimensional environment for culturing brain cells. By coating the bottom of the dish with a nontoxic repellent, he coerced neural progenitor cells to float freely in their nutrient broth, rather than hug the bottom of the dish as is their wont. Thus suspended, the cells proliferated, differentiated and clustered into tiny balls. These almost perfectly round clusters of cells continued to grow, differentiating further and eventually approaching one-sixth of an inch in diameter and perhaps a million cells apiece, forming brain-region-specific spheroids.
“The 3-D experiments were more of a game in the beginning,” Pasca says. “They started as an exploration of how much self-organization one can see in a dish after coaxing stem cells toward a neural fate. I was new to developmental neurobiology and fascinated by how cells assemble to form such complex structures as the mammalian central nervous system.”
Anca Pasca, who was doing her pediatrics residency at Stanford during that time, would often come to the lab to do experiments on her own. “After we moved here in 2009, Sergiu began spending a lot of time in the lab,” she says. “We had no friends and no family here, and I was getting bored staying home studying for my medical boards. We started wondering, what if we let these neural progenitor cells just float, suspended, in the dish instead of letting them attach?”
“She thought this could be a big thing,” says Sergiu Pasca, “and she continued to work on optimizing the culture medium and characterizing the little spheres.” Initially, Pasca and his colleagues produced brain balls by seeding their 3-D cultures with neural progenitor cells they generated from iPS cells grown in standard 2-D cultures. (Pasca now refers to this as the “2.5-D” method.) But eventually they learned how to proceed entirely in three dimensions, starting with actual iPS cells they then guided, in 3-D culture, through the neural-progenitor phase and then all the way to full-fledged, all-brain-and-only-brain brain-ball status.
Pasca’s brain balls can remain viable for up to two years or longer in culture — a record duration, long enough not only for the neurons they contain to mature thoroughly, but for their resident neural progenitor cells to spawn viable astrocytes, too. That, in turn, makes it possible for the neurons to form functioning synapses and complex circuits.
By mid-2014, Pasca had an assistant professorship in a tenure-track job and his own lab at Stanford. He also had multiple rounds of culture-medium optimization and brain ball characterization under his belt. The neurons in the brain balls acted, for all practical purposes, like the ones in a living brain. Their synapses were in working order, allowing neurons to form complex circuits through which they could talk to one another. These circuits closely approximated the real architecture of the brain region they’d been coaxed to mimic.
“I found myself wondering why people hadn’t tried this before,” says Pasca. In fact, others have used 3-D tactics to produce cultures enriched for brain cells, but they contained haphazard combinations of cells from other tissues. They also lacked the Pasca brain balls’ astounding brain-region mimicry.
Patience yields results
The key missing ingredient: patience. While others simply allowed their cell cultures to grow in a dish in a hands-off, nondirected fashion, the Pascas and their lab mates coaxed and coached their brain balls along desired developmental pathways by applying various combinations of small-molecule nutrients, trial-and-error style, iteration after iteration.
The Pasca group published its findings in a paper in Nature Methods in May 2015. One of the most amazing things about their brain balls was that, with not much chemical guidance, they tended to take on a default structure that’s a facsimile of the most evolutionarily advanced part of the brain: the human cerebral cortex, with all six layers you find in a living human brain.
The types and amounts of various proteins that each layer’s neurons were manufacturing mirrored those of the equivalent layer in an actual human cortex. And those neurons were alive and kicking: They sported ship-shape synapses, displayed spontaneous activity, fired in networked synchrony and were surrounded, as in real life, by their BFFs: happy, healthy astrocytes. (Astrocytes always pop up spontaneously in brain balls if you culture them long enough.)
“It’s amazing that these cells already self-organize and know what they need to do,” Pasca marvels. The iPS cells Pasca’s team uses as starter seeds for brain balls are generated from skin cells, so researchers can produce brain balls whose cells are genetically identical to the brain cells in the person whose skin is used. That makes brain balls powerful tools for studying any neurological disorder with a genetic component.
“Early on, I didn’t really believe this was going to work,” he says. “But Anca continued to help me optimize the protocols for the culture system until it did.”
Anca Pasca was a lead author of her husband’s 2015 paper, and he credits her for much of the hands-on work that helped to turn those “Saturday experiments” to a front-burner project. Her co-lead author, Steven Sloan, PhD, is an MD student who did his graduate work in the laboratory of the late Ben Barres, MD, PhD. On acquiring his PhD, Sloan has continued his research in the Pasca lab.
Throughout his postdoc period and beyond, Sergiu Pasca also was encouraged in his 3-D culture push by Barres, whom he calls his “second mentor.” Barres, who died in December 2017, was a professor of neurobiology, of developmental biology and of neurology and neurological sciences. Much of what’s known about astrocytes stems from Barres’ decades of research. The two notoriously late-night workers would engage in conversational collisions in the hallway or stop into each other’s adjacent offices when they saw the light on.
Later, when Pasca moved his lab to a new location, they would often exchange emails or text messages until early morning. “He really believed this could be done when nobody else did,” Pasca says. Barres also gave him an ultimatum: “He’d warn me that if my cultures weren’t producing astrocytes, they were crap.”
“Sergiu has accomplished something quite magnificent,” said Barres in an interview before his death. “The power and promise of this method is extraordinary. You can watch all kinds of brain diseases developing in a dish.”

A brain ball isn’t limited by fate to become a little cortex Mini-Me. With the right factors supplied at the right time, each can take on (neuroanatomically speaking) a personality of its own. This means researchers can make brain balls that mimic two different brain regions containing different kinds of neurons, fuse the balls together and see how they talk to each other. Pasca’s group has done just that, showing in an April 2017 paper in Nature that when two brain balls representing different brain regions are brought into contact, they can fuse and forge complex neuronal connections.
When a brain ball meets a brain ball
In real brains, excitatory neurons secrete a substance whose arrival at one of its receptors on the next neuron in a relay increases the likelihood of that next neuron firing off an impulse. During fetal development, the cerebral cortex’s excitatory neurons are eventually joined by inhibitory neurons that originate in an underlying region of the developing forebrain. Those inhibitory cells migrate several millimeters to the cortex, where they interlace with excitatory cells to form circuits that drive the brain’s advanced cognitive activities — and in which an excitation-to-inhibition imbalance can lead to epilepsy and has been suspected to play a role in autism. But no one had been able to watch this happen to human cells in real time in a dish, or anywhere else.
In the study, Pasca’s team cultured two separate batches of human brain balls generated from a healthy subject’s skin. They bathed one batch in a medium that fostered cerebral cortexlike brain balls containing excitatory neurons. They placed the other batch in dishes whose nutrient broth steered balls toward resembling the underlying brain region where inhibitory neurons originate. Then, the investigators juxtaposed the two distinct brain ball types in the same dish.
So, what happened in the petri dish when excitatory and inhibitory brain balls touched? Within a few days, the balls fused, and the researchers could watch inhibitory neurons from one migrating into the other, excitatory-neuron-rich one.
On reaching their destination, the inhibitory neurons sprouted dendrites — the foliagelike “tails” that neurons use for receiving inputs from other neurons — and hooked up with the excitatory neurons. The two neuronal types formed functional, mutually signaling circuits.
Then the investigators generated both types of brain balls from skin samples of patients with the autism-related Timothy syndrome, fused them and watched. What they saw was this: The inhibitory neurons appeared to develop normally at first, but migrated in a markedly inefficient way. Pasca’s team showed that two different drugs could restore normal migration and, ultimately, normal signaling properties to the Timothy-syndrome-derived inhibitory neurons.
Running with the spheroids
Anca Pasca has taken a two-year sabbatical from her clinical work to concentrate on using the brain balls to study hypoxia, an oxygen deficiency in the brains of babies born before their lungs have matured sufficiently.
“Technology now allows us to save babies as early as 22 to 23 weeks of gestation,” she says. “But up to 90 percent of these saved preemies will wind up with significant neurodevelopmental impairment. We see a lot of hypoxia in the NICU [neonatal intensive care unit]. We think it’s one of the most important causes for abnormal brain development ranging from mild learning deficiencies to cerebral palsy.”
“Thanks to three-dimensional culture, we now have a way of mimicking developing brains’ hypoxia exposure in a dish and I can study them. Once I understand what’s happening to them, the next step is to come up with a drug to prevent this from happening.”
Pasca’s brain-ball-generating method has been reproduced, and put to work, in numerous other labs, and not just to study early development.
“Pasca’s technique has become the standard method for generating neural spheroids in three dimensions,” says Boston University School of Medicine research associate professor Weiming Xia, PhD. In a study published in 2016 in PLOS One, Xia and his colleagues compared brain balls à la Pasca with old-hat 2-D neuronal cultures as vehicles for screening a couple of experimental drugs targeting Alzheimer’s disease.
The scientists found that the drugs — both of which work by reducing the production of a substance called A-beta that aggregates into gummy plaques characteristic of the disorder — showed less potency in brain balls than in 2-D neuronal cultures. That means a lot of expensive follow-up after a positive result using the 2-D assay could turn out to be a waste of time.
The reason for the drugs’ reduced efficacy in brain balls, Xia says, is that they had a hard time getting inside the brain balls. Because brain balls approximate the brain far more closely than flat sheets of neurons at the bottom of a petri dish, they probably provide more meaningful results, he says, and could help steer research to drugs that do find ways to penetrate and permeate the spheroids.
And in Pasca’s lab, the team has made brain balls resembling other important brain regions such as the midbrain structure called the striatum, a key component for facilitating movement and the brain’s reward system. The hope is to fuse the striatal balls with cortical balls and try to generate, in a dish, a nerve tract running between the striatum and the cortex that’s involved in movement disorders such as Parkinson’s and Huntington’s diseases as well as psychiatric conditions such as obsessive-compulsive and attention-deficit hyperactivity disorders. And they’re trying to replicate the spinal cord and a deep-brain structure called the thalamus — a relay station for sensory inputs.
Thalamic brain balls will be particularly useful to John Huguenard, PhD, professor of neurobiology and of neurosurgery and one of the co-authors of Pasca’s 2015 paper, who is particularly interested in the circuits by which the thalamus communicates with the cortex, and vice versa.
“Genetic variants are estimated to account for up to 50 percent of seizure susceptibility,” says Huguenard. If Pasca’s brain ball-fusion results in the recapitulation of the nerve tracts connecting the two regions, Huguenard can study at close range the effects of genetic insults that induce a predisposition to epilepsy in a developing brain.
Pushing the boundaries
These balls of cells floating in a petri dish lack some crucial features of functioning brains — sensory input, for example, and connections to muscles, the immune system and a circulatory system. But Pasca thinks some of these problems will find solutions. Transplanting human brain balls into rodent brains could result in their tapping into the animals’ circulatory systems. He hopes to implant such fusion products, or just single brain balls, into a mouse, placing a patient-derived construct on one side of the mouse’s brain and another one derived from a healthy person on the other, then comparing how the two different brain balls operate in that living environment.
Pasca acknowledges the need for caution. “This type of transplant experiment requires approval like all of our experiments, but we are also actively engaged in conversations with ethicists and other scientists in the field about the best way to move forward.”
He’s part of a group working with the National Institutes of Health to create guidelines for the quickly advancing field. Where would he draw the line? “Primates. I would not want to transplant human brain cells into a nonhuman primate without a strong rationale.”
And interest continues to build. Pasca organized a December 2017 inaugural meeting focusing on three-dimensional modeling of the human brain. The meeting, held at Cold Spring Harbor Laboratory in New York, drew 130 participants from around the world, and he’s co-chairing a weeklong course in Italy this summer. Stanford has licensed the technology to a Vancouver-based company, STEMCELL Technologies, which plans to release brain ball culture kits that could speed researchers’ adoption of Pasca’s advance.
Meanwhile, Pasca’s lab now boasts almost as many different cultures as the United Nations. “We have hundreds of iPS cell lines obtained from patients with various psychiatric disorders,” he says. While planetary peace may be a distant development, learning how a living brain develops is a dream that’s rapidly coming true.

