To hear again
Birds regrow damaged inner ear cells. Why can’t we?
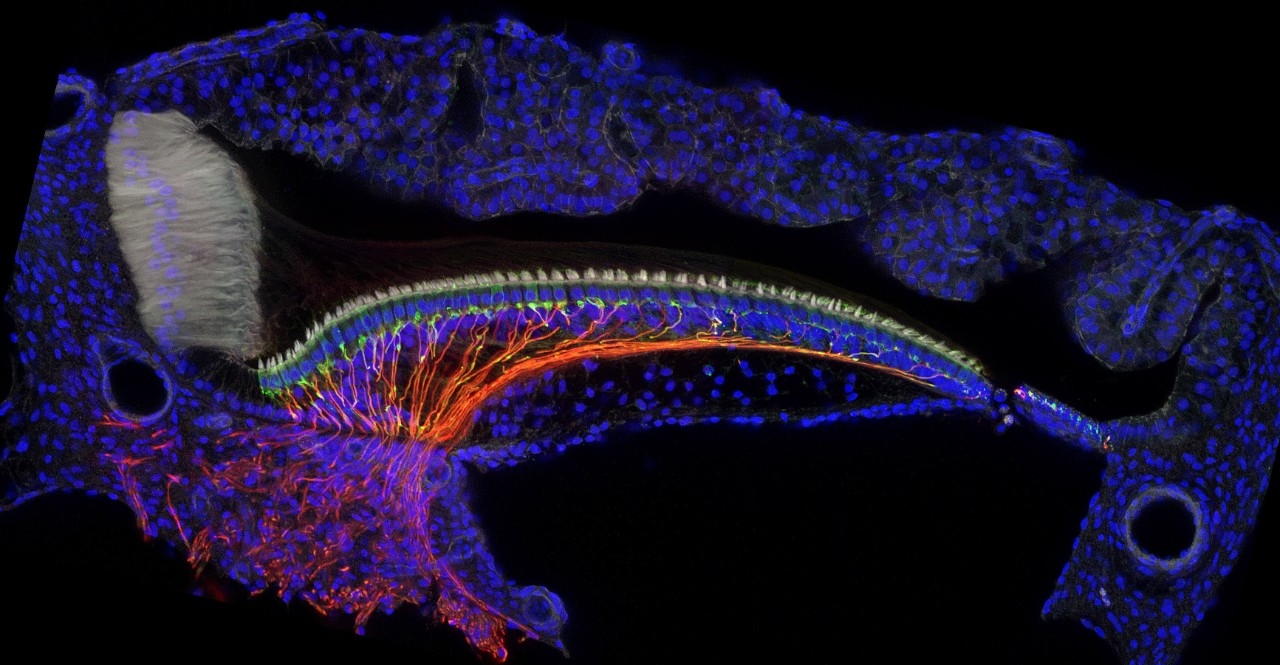
Hunkered over her computer, Amanda Janesick, PhD, a postdoctoral scholar studying the biology of hearing, points to a series of microscopic images on the screen. The time-lapsed cross sections of a chicken’s inner ear show multitudes of hearing cells dying off and multitudes of new ones replacing them.
This is interesting to Janesick — and many others concerned about hearing loss — because these cells function similarly in humans, with an important difference. In birds and mammals, thousands of these hearing cells, called hair cells because of their hairlike cilia, are hidden deep inside the fluid-filled cochlea, a tiny, bony structure.
For a human or a chicken to hear, the swaying cilia on the hair cells sense sound vibrations from the outside world. The vibrations cause them to deflect, which opens pores or channels that allow electrical signals to be sent via the auditory nerve to the brain, which interprets the signals as sound.
Here’s the key difference: If these hair cells are destroyed in humans, they lie around like flattened wheat after a rainstorm, permanently powerless to help us hear. The cells die for various reasons, including injuries, aging and loud noises. And no new cells replace them.
But in birds, amphibians and reptiles these cells grow back. So birds, unlike humans, never go permanently deaf.
Scientists want to make humans more like birds.
“The goal is to follow the trail of these regenerating cells in birds. Then we have to figure out why this trail is not working in mammals.”
“The goal is to follow the trail of these regenerating cells in birds,” says Stefan Heller, PhD, professor of otolaryngology-head and neck surgery, who heads the lab where Janesick is working. “Then we have to figure out why this trail is not working in mammals. What are the missing parts? Maybe we will discover one critical trigger and we can cleverly fix this trigger in mammals.”
New research in Heller’s lab could finally lead to cures for hearing loss in humans by helping scientists understand the stepwise process by which individual cells, rather than entire groups of cells, regenerate into functioning hair cells.
“A fundamental piece is still missing,” says Heller, who for the past six years has been using new tools to figure out how thousands of unspecialized cells, such as stem cells or progenitor cells, transform into hair cells. “We don’t know how regeneration is being orchestrated. When a hair cell is dying, what signal is this hair cell sending out to a neighboring cell, and what activates these neighbors to start the regenerative process?”
This search for biological discoveries in hearing loss has drawn the attention of biotech companies attempting to develop new products based on research from scientists like Heller. (Heller is a scientific adviser for one of those companies, Inception 3 Inc.)
Witnessing new cells arise
In the chicken cochlea images in Heller’s lab, Janesick points to groups of supporting cells that are visible beneath the rows of hair cells — the progenitor cells. It appears that, as time passes, these cells turn into, or create, new hair cells.
Though the chicken cochlea is roughly banana-shaped and the human version looks like a snail shell, both are similarly filled with thousands of hair cells — 10,000 in chickens, 15,000 in humans. To witness how hair cells regenerate, Janesick first injected an antibiotic known to be toxic to hair cells directly into a chicken’s cochlea. Her images show the progression as the drug kills off the hair cells within 24 hours, and then how multitudes of supporting cells grow rows of healthy, new, cilia-sprouting hair cells during the subsequent two weeks.
“We need to learn a lot more about those supporting cells,” says Janesick, a developmental biologist whose mild hearing loss helped spur her interest in working in Heller’s lab. “What are they, what do they do, how do they communicate with each other? And, ultimately, why can these cells regenerate hair cells when similar supporting cells in mammals appear to be nothing but a dead end?”
The genesis of hearing loss research
Heller’s interest in hair cells goes back to 1994, when he had just received a PhD from the Johannes Gutenberg University in Mainz, Germany, and was seeking an interesting project.
“I was literally looking through the book Principles of Neuroscience, and I found a chapter on hearing and hair cells,” Heller says. “The ear appeared so complex, and I knew pretty much nothing about how it works. It just seemed that this is an area of biology that still had open questions, lots of challenges. When people started asking me, ‘Why in the world would you choose a topic like that?’ — I realized that I had found my place.”
By 2002, Heller had gained international attention for being the first to identify stem cells — undifferentiated cells that can develop into various specialized cells — that reside within the inner ear. Over the next six years, his lab successfully developed mouse cells from these stem cells that looked and acted just like the animal’s inner-ear hair cells. In 2010, they published a study in Cell reporting these results as a major advancement.
The researchers hoped to perfect the recipe in human ear hair cells so they could generate the millions of cells that would be needed for transplantation into the ear. But they couldn’t get the cells to do their bidding.
“The early human ear cells we generated were just not stable enough. No one could figure out how to keep the cells on the desired inner ear trajectory; they always became something else. That was the end of the line for us,” Heller says.
That’s when his lab turned to technology to help understand how individual progenitor cells develop into hair cells in birds.
“It might not be a perfect fix. It might not fully cure deafness. But it may turn a hard-of-hearing person into a better-hearing person. That is the first step we have to take.”
We figured out that we had to get to the point where we understand what the thousands of individual cells are telling each other, and not how the organ is working as a whole, which is what we had been doing for many years.”
A decade ago, measuring such complex and extensive information would have been impossible, but technological advances enable researchers to separate individual cells and measure their transcriptome — that small percentage of the genetic code that is transcribed into RNA molecules. By studying transcriptomes, researchers hope to determine when and where genes are turned on or off in various types of cells and tissues.
“Technology has really turned the tables for us,” Heller says.
About six years ago, Heller’s lab adopted a technique, called single cell transcriptome analysis, invented in part in the lab of Stephen Quake, PhD, a professor of bioengineering and of applied physics. The process uses high-tech tools to decode the genetic instructions that enable cells to differentiate into other cells.
“We are analyzing all the genes expressed in individual cells during regeneration,” Heller says. “We just published a paper describing how the chicken utilizes two parallel regenerative processes. We need to figure them out, how they change over time. The cells are mixed up together and when we isolate the cells, we lose critical information, such as their precise location in the organ. Currently there are no informatics tools that allow us to reconstruct that so we’re creating our own.”
Heller’s lab has been hosting bioinformatics experts who are working on algorithms to describe how individual cells respond to regenerative or developmental triggers, and how they make decisions to develop into different mature cell types.
“We have really had to focus on generating the right tools with the intent of applying this technology, which I think is going to revolutionize the whole field,” Heller says. “We also plan to eventually apply this to our stem cell work.”
Heller is cautiously optimistic about the ability to develop hearing loss treatments. Work in genetic therapy and drug development to protect against the hearing loss side effects of aminoglycosides, a widely prescribed class of antibiotics, may be a bit closer to fruition, he says. But in general, inner ear hair cell researchers are in this for the long haul.
“In this field, there is no magical cure,” Heller says. “It will be a process. For example, if we discover that ‘trigger’ in chickens and fix it in mammals, we may be able to trigger something like an imperfect regeneration of hair cells in humans. It might not be a perfect fix. It might not fully cure deafness. But it may turn a hard-of-hearing person into a better-hearing person. That is the first step we have to take.”

