The cell whisperer
An engineer sways tissue samples to behave in the lab like they would inside us
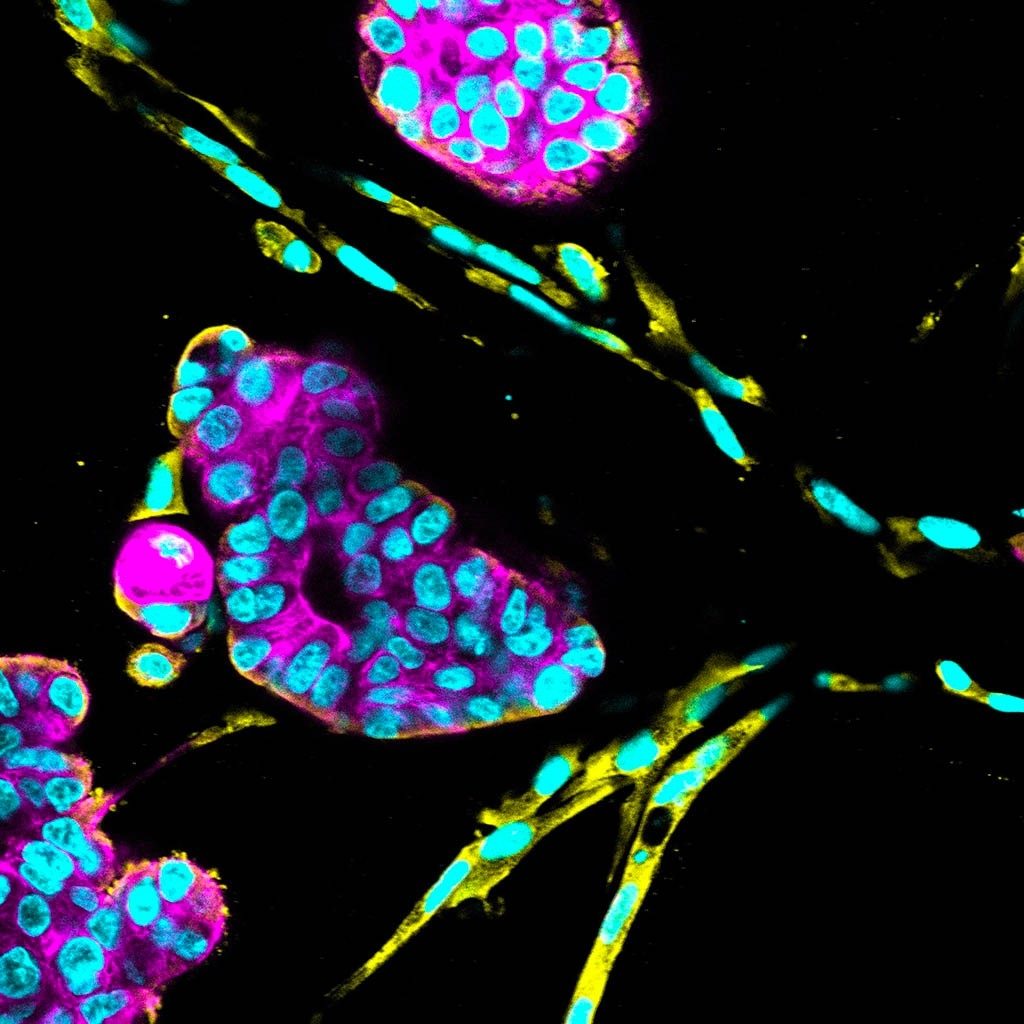
To the untrained eye, the globs of cells about the same size as a dust speck do not look like much, certainly not miniatures of the full-sized organs they represent. But these organoids are part of the latest revolution that could open up drug discovery and personalized medicine.
Until the 1950s, cell biologists struggled to keep human cells alive outside a person. But in 1951, Henrietta Lacks’ aggressive cervical cancer cells, the first human cells that scientists were able to duplicate and grow in the lab, changed that. Researchers used these cells — named HeLa — to conduct some of modern medicine’s landmark research studies and to learn how to immortalize many other types of cells.
Even with these advances, cell culture technology commonly used today remains inadequate for answering some important questions about the lives of cells. That’s because the cells’ habitats are largely two-dimensional.
“We culture them on a petri dish. It’s just a flat dish,” explained Sarah Heilshorn, PhD, associate professor of materials science and engineering, sketching a flat surface with her hands. “They’re isolated little cells, and they’re not doing anything like what they do in our bodies.”
Biologists now want to coax cells into systems that more closely resemble human tissue to gain better insights into human development and disease. In the past decade, they have started growing cells in three-dimensional structures.
Heilshorn, a 2009 National Institutes of Health New Innovator Award winner, is helping scientists build these structures to aid medical research at Stanford and elsewhere. Her specialty is developing new materials — known as scaffolds — to support the developing organoids. Though her work in the lab was on hold at the beginning of the COVID-19 pandemic, she restarted experiments in June.
A race to model organs
In 2009, Calvin Kuo, MD, PhD, a professor of medicine at Stanford, and Hans Clevers, MD, PhD, at the Hubrecht Institute in Utrecht, the Netherlands, both reported that they successfully grew mouse intestine as organoids — a collection of cultured cells that emulate the natural architecture of the intestine within the body.
The achievement kicked off a race to develop organoids that model other major organs, including the liver, breast, eye and brain. Most laboratories grew organoids on a commercially available protein mixture called Matrigel, which is a gel derived from mouse tumors.
But Matrigel is not perfect. This stew contains 2,000 or so different compounds, and there’s a lot of variability among batches, which creates problems, explained Ruby Dewi, a biology researcher in Heilshorn’s group.
So a few researchers, including Heilshorn, have started developing artificial gels, called hydrogels. Importantly, they know exactly what they’re putting into these hydrogels. “We try to make materials that in some way mimic those that are found in our own bodies,” she explained.
Despite the broad range of disciplines in which she has worked — she began as a chemical engineer but went on to study chemistry, physics, bioengineering, and cell biology — Heilshorn sees herself as an engineer. Engineers, said Heilshorn, are “reductionists by nature.”
“We want to make as simplified a model as possible to capture some biological phenomenon that we think is important,” she said.
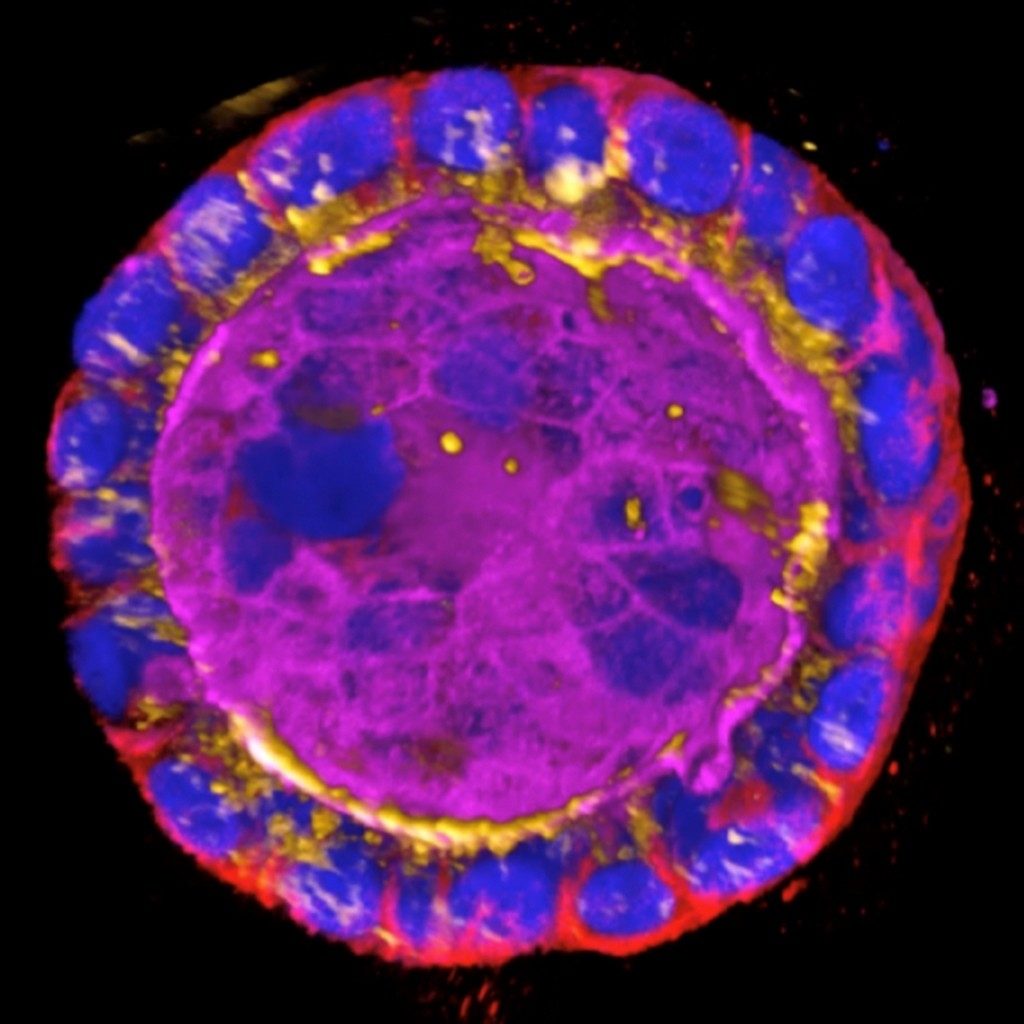
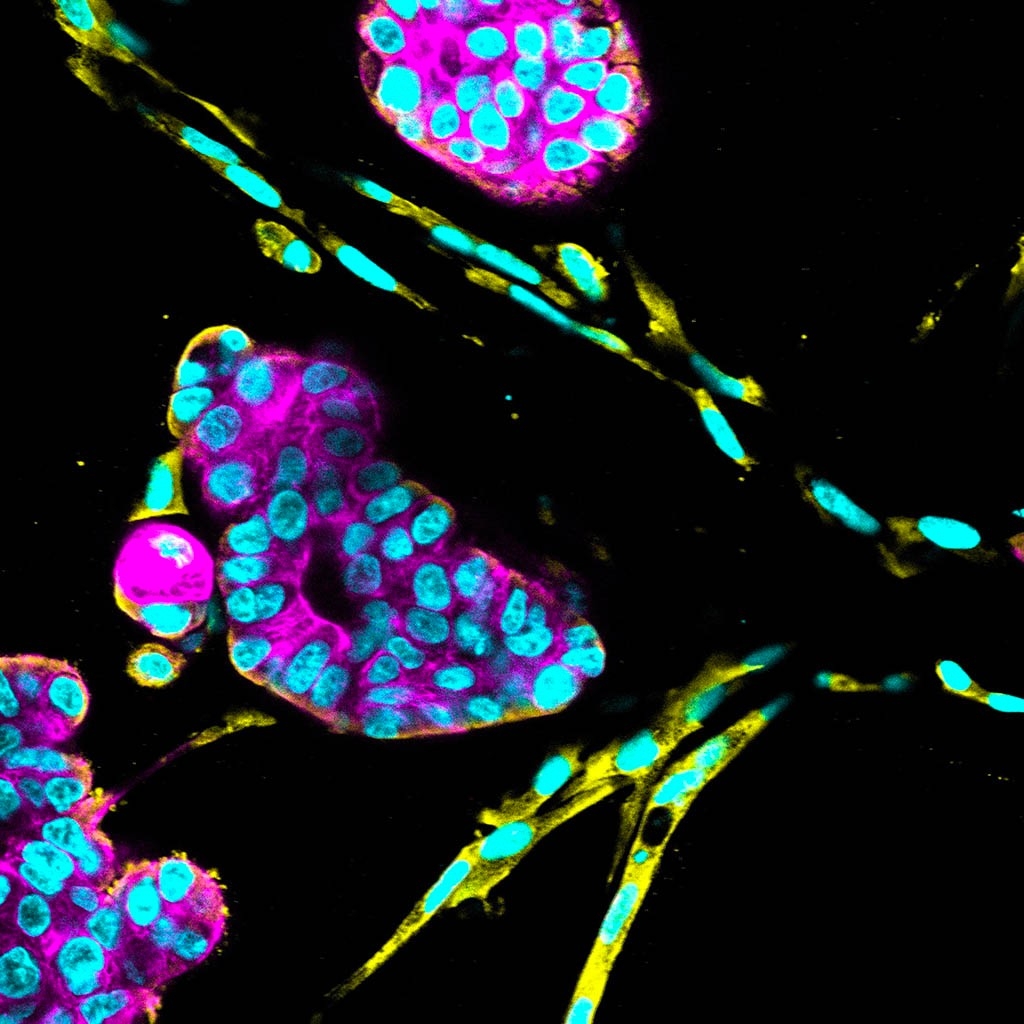
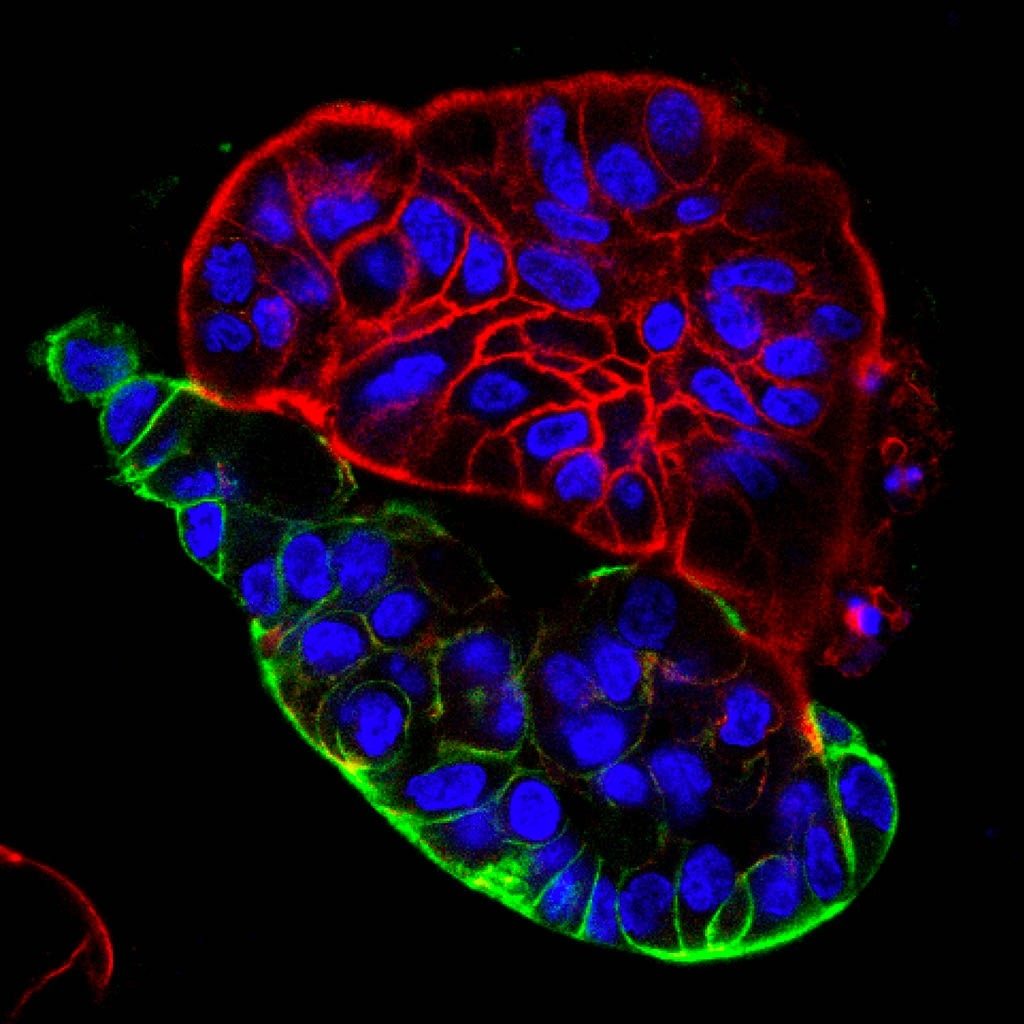
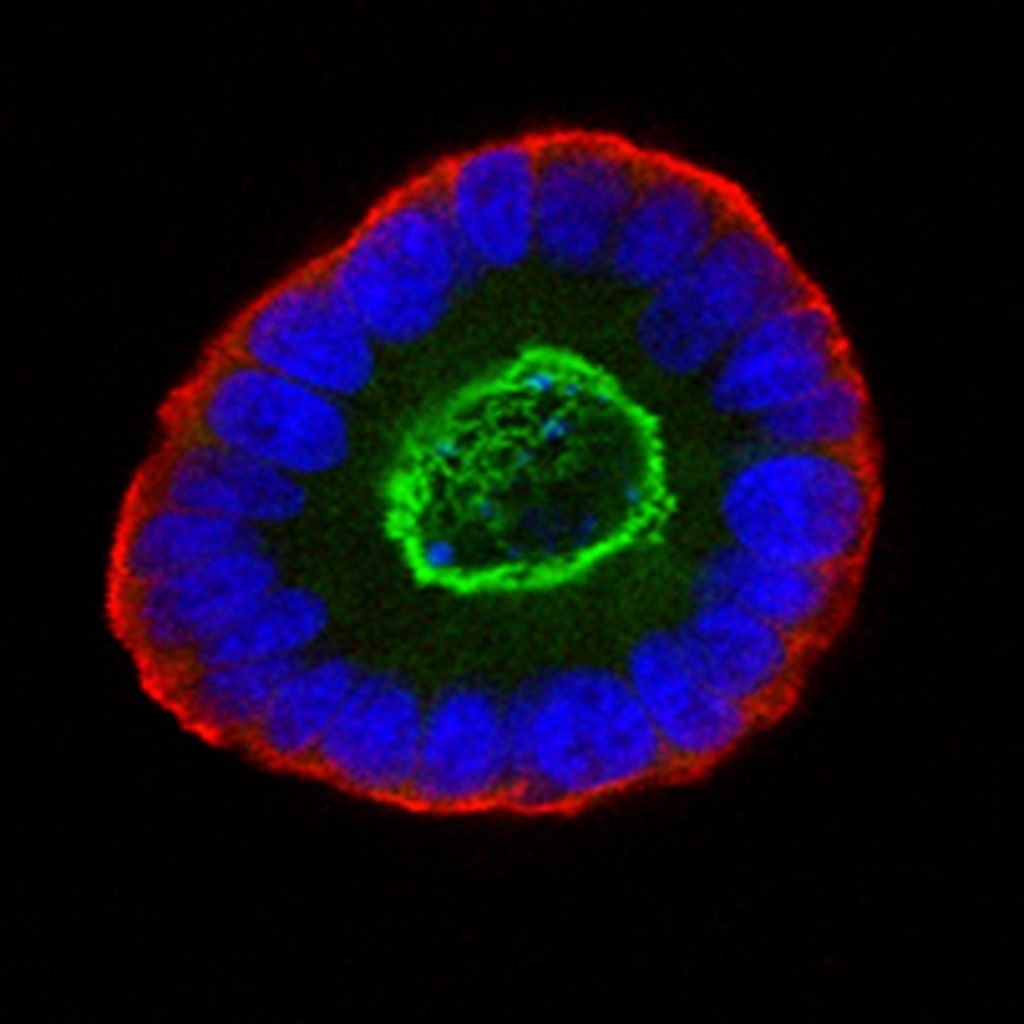
The tricky business of growing stem cells
Many medical researchers are especially interested in growing stem cells because their stemness — or ability to self-renew and form all different kinds of cells — makes them invaluable for understanding how tissues develop.
But growing stem cells is tricky. Researchers want to culture stem cells in their undifferentiated, or unchanged, state. If the cells begin to differentiate into specialized cells, such as brain or lung cells, they lose their stemness, and scientists have difficulty coaxing them into other desired cell types.
University of Wisconsin-Madison researchers were the first, two decades ago, to extract human embryonic stem cells that were kept alive in the laboratory. Scientific advances since have made it easier to maintain stemness in 2D cultures than in a 3D environment.
These 2D cultures, however, are quite different from the environment found inside the body where the stem cells would naturally differentiate, which means the cells inside those cultures will behave differently. The ability to develop 3D stem cell cultures, though difficult to achieve, would be a great advance for researchers.
Fine-tuning gels to fine-tune cells
To better understand how the properties of the 3D material influence cell stemness, Heilshorn and her collaborators designed a family of 3D hydrogels from proteins with tunable mechanical properties.
In other words, the researchers could make small changes to the gel’s properties — endowing them with more flexibility, for example — and then systematically test how the change affected a specific stem cell population.
Past studies had earmarked matrix stiffness — basically, the rigidity of the gel — as an important factor in maintaining cell stemness.
But Heilshorn and her research collaborators found that varying degrees of stiffness had little effect on stemness for the neural stem cells they examined. Instead, cells tended to retain their stemness better in the gels that more easily break down and remodel. The team published their results in December 2017 in Nature Materials.
When working with biomaterials, changing one variable generally causes another property to change as well, making it difficult to observe the effects of a single variable’s change. But Heilshorn’s approach allowed the team to independently study stiffness and matrix remodeling.
As it turns out, physical contact between the neural stem cells helps them retain stemness, explained Heilshorn. The researchers found that the neural stem cells divided and made copies of themselves only after they could touch each other. If the properties of the gel prevented cell connections, the cells couldn’t grow and divide as they normally do in the body.
“The engineering of the material allowed us to identify something about the biology,” said Heilshorn.
Nurturing organoids to sprout and grow
Curiosity has driven Heilshorn since childhood. “I was one of those kids who’d ask ‘Why?’ like a thousand times every day,” she said.
Collaborations with Stanford Medicine researchers allow her to satisfy her curiosity about a wide range of medical matters. Early research with Kuo, a pioneer in developing new organoid culture methods, sparked her interest in organoids. In their latest projects, they are growing intestinal organoids and cancer organoids, both from patient biopsies.
“You just drop it in and it needs to sprout and grow — it’s actually like a plant. Sarah’s trying to come up with better soil, basically.”
Calvin Kuo, MD, PhD, a professor of medicine
Organoids have typically been grown underneath the tissue culture scaffold, but Kuo uses an air-liquid interface that directly exposes the organoids to air. The technology allows him to grow larger fragments of tissues, which is helpful for studying diseased tissue.
He’s been using Heilshorn’s artificial materials to find the conditions that allow the organoids to grow even better.
The organoid is the seed and the matrix is the soil, said Kuo. “You just drop it in and it needs to sprout and grow — it’s actually like a plant,” he explained. “Sarah’s trying to come up with better soil, basically.”
Once you can grow the organoids, you can study them.
One area of great interest is growing tumor organoids from a patient biopsy and testing out potential drugs.
“We are attempting to expose those tumor biopsies to many different treatments in the lab so that we can possibly suggest, or maybe even ultimately predict, to which treatments a patient’s cancer would best respond,” said Kuo. “That’s personalized medicine.”
Such work with organoids isn’t limited to cancer. Kuo is looking at such genetic disorders as cystic fibrosis and such autoimmune conditions as celiac disease. “There’s just any number of possibilities,” he said.
Tackling ovarian cancer
One of those possibilities is taking shape elsewhere on campus, where Heilshorn is using her biomaterials expertise to tackle ovarian cancer in collaboration with Erinn Rankin, PhD, an assistant professor of radiation oncology and of obstetrics and gynecology, and Oliver Dorigo, MD, PhD, a surgeon and associate professor of obstetrics and gynecology.
Ovarian cancer is the fifth-leading cause of cancer-related deaths for American women, according to the American Cancer Society. The big problem, explained Rankin, is that by the time most women are diagnosed, the disease has already escalated.
“It’s almost like a cottage cheese, spread throughout your abdomen,” she said.
“That advanced disease has a 30% chance for survival compared with 90% survival in early stage disease,” added Dorigo, who operates on women with ovarian cancer.
Most patients respond well to their first chemotherapy, but the disease recurs, said Rankin. And because their cancer is so advanced and widespread, it becomes resistant to subsequent rounds of treatment. Heilshorn’s research goal is to help figure out why this happens.
With samples donated by Dorigo’s patients, the team gets a snapshot of the different types of cells. Studying only the ovarian cancer cells, as is commonly done, is limiting, because the different cell types in tumor microenvironments communicate with each other.
“We’re really excited, because we’ve identified a new cellular component of the tumor microenvironment that promotes chemo resistance.”
Erinn Rankin, PhD, an assistant professor of radiation oncology and of obstetrics and gynecology
For example, the cells surrounding a tumor respond to the tumor’s cells by producing extra collagen, which becomes stiff and fibrous, said Heilshorn.
Not only can her team characterize the material properties of the tissue, as well as its variety of cell types, they can also use that information to engineer 3D synthetic versions of the cancer matrix — their own ovarian cancer organoids — which they can study further.
Though the researchers are approaching the end of their two-year grant from Stanford Bio-X to pursue this line of inquiry, they plan to continue their collaboration, and they have new grant applications pending. Meanwhile, they are preparing to submit a manuscript for publication in a peer-reviewed journal.
“We’re really excited, because we’ve identified a new cellular component of the tumor microenvironment that promotes chemo resistance,” said Rankin.
If they can figure out what makes the cancer cells become resistant, they can develop more effective therapies for women with ovarian cancer.
What’s more, the models created by the team could be useful for identifying new biomarkers to aid early detection of the disease, said Dorigo.
“To me, the real beauty of being able to create a living sample of a patient’s tumor is that you can study it from every different angle and in this exquisite detail that you couldn’t if it were actually in the patient,” said Heilshorn.
“My hope is that by doing that, we’ll be able to really answer some more fundamental questions about the biology of cancer.”
Contact Laura Shields at medmag@stanford.edu. Portions of this article originally appeared in UC-Santa Cruz Science Notes in 2018.
