Here come the assembloids
A new type of brain model reveals the organ’s workings in unparalleled detail, providing insights into development and disease

Sergiu Pasca was nervous. Let us count the reasons why.
Because of a pandemic-enforced hiatus, he hadn’t spoken publicly — and indeed, like many of us, had greatly curtailed public contact — for two years.
Now, on the morning of April 12, Pasca, an associate professor of psychiatry and behavioral sciences at Stanford Medicine, was giving a TED talk in Vancouver before an audience of about 1,500. Hectobillionaire Bill Gates had preceded him by a couple of speakers. And he’d been instructed to establish deep eye contact with the strangers in the front row. (That really freaked him out, he said.)
He may have been making the audience a tad nervous too. He was telling them about a discovery guaranteed to raise eyebrows and blood pressure: how to turn anyone’s skin into a replica of a small portion, or portions, of that person’s brain, thriving and growing inside a lab dish.
Pasca, the Bonnie Uytengsu and Family Director of the Stanford Brain Organogenesis Program, assigns a fair share of credit for this feat to the brain. “The human brain largely builds itself,” he said. “It comes with its own assembly instructions.”
Which is fortunate. If, as rumored, the brain really is the most complicated thing in the universe, we’re awful lucky we don’t have to build our own. Of course, if you’re trying to get one to grow in a dish, you are going to have to do some prompting.
You can’t just pour in some skin cells, add water and expect the little blob to respond with a hearty, “Thanks, I’ll take it from here.” You’ll need to fold in a few key ingredients to coax the component parts of that most motley of all organs, the brain, into existence and cooperation.
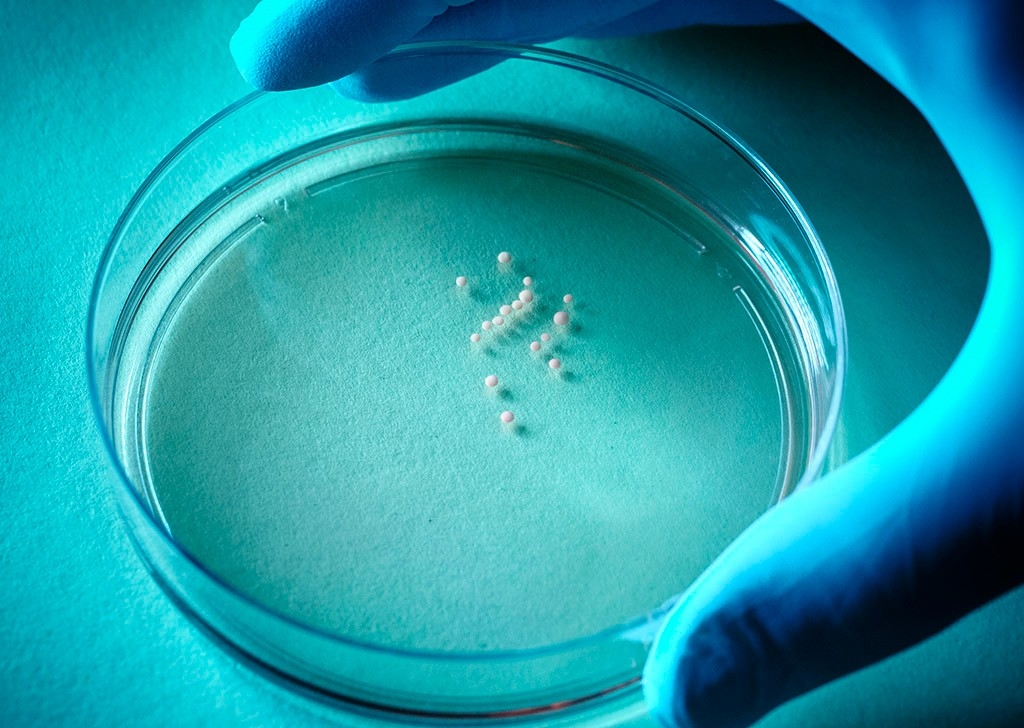
Pasca gets credit for figuring out the ingredients and conditions that encourage distinct parts of the brain to self-assemble as clusters called organoids in laboratory glassware.
Now he has taken it a step further, coaxing the organoids to merge into agglomerations he calls “assembloids,” in which cells migrate or extend projections from one organoid to the next, establish connections and build circuits that are credible, accessible working models of their mostly inaccessible real-world counterparts.
That lets researchers get action close-ups of the nearest thing to a living human brain that’s ever come along without needing brain tissue from anyone living or deceased.
They can learn how a healthy brain develops in a fetus or maybe even a newborn. And they can detail the deficiencies that derail brain function and cause neurodevelopmental disorders such as autism, schizophrenia and epilepsy.
Pasca arrived at Stanford Medicine in 2009, an MD from Romania with little training as a researcher or, for that matter, fluency in English. Now here he was, rubbing elbows with the likes of Gates, Elon Musk and more, and broadly considered the pioneer of an approach that’s since been adopted by well over a hundred labs around the world, including those of several Stanford collaborators.
Stanford has licensed the methodology to a private company, Stem Cell Technology Inc. If you’re a neuroscientist and you want to study the workings of one of the brain regions — or a combination of them — Pasca’s lab has figured out how to make, you can buy a kit to do it yourself. You get starter parts plus reagents plus instructions.
A new era of brain research — what Pasca likes to call “molecular psychiatry” — has dawned. It promises new revelations about our most mysterious organ’s inner workings and once-opaque development.
It offers the prospect of living laboratories in which to test pharmacological and electrophysiological methods of curing nervous-system disorders that arise during early development or from infections and injury later on.
And it will catalyze not only comprehension and cures but also conundrums, as the products of Pasca’s innovation inch forward to ever-increasing complexity, and start to resemble parts of our own brains in ways that are bound to concern us.
His TED talk — 13 years of research condensed into a 14-minute presentation — was interrupted more than once by applause.
The rise of the assembloids
Here’s a whirlwind recap of what took place during that 13-year period and the three-year gestation that preceded it.
In August 2006, Japanese researcher Shinya Yamanaka, MD, PhD, published a paper in Cell chronicling his lab’s successful transformation of skin cells into “induced pluripotent stem cells,” or iPS cells — stem cells that are capable of multiplying in a dish and, almost magically, of potentially differentiating into all the 200-plus types of cells in the body.
Cut to three years later. Pasca, new to the United States, began a postdoctoral fellowship in the lab of Ricardo Dolmetsch, PhD, then an assistant professor of neurobiology at Stanford Medicine.
Starting with iPS cells, Dolmetsch and Pasca generated neurons derived from the skin cells of a young patient with a rare genetic disorder called Timothy syndrome, which predisposes people to autism, epilepsy and cardiac dysfunction.
Plated on plastic in a lab dish, the neurons grew in culture and could be used to study the disorder on a patient-by-patient basis, without a brain biopsy. The researchers published these results in a cover article in Nature Medicine in 2011.

But the neurons were a little sickly, didn’t populate the dish very thickly and died too quickly to form working circuits. However, in a breakthrough experiment in 2011, Pasca found that suspending stem cells in a lab flask could induce them to multiply and form spherical clumps.
He could then guide those clumps of neurons to differentiate and recapitulate, in three dimensions, some of the features of the brain’s outermost and most evolutionarily advanced section: the cerebral cortex.
These organoids — small masses, maybe one-sixth of an inch across and composed of about a million cells — thrive in culture, some of them for more than 800 days. That’s long enough for some very slow-maturing brain cells to make their appearance, enabling researchers to see things happen that would have been missed using any other experimental system.
For example, some brain cell types spontaneously undergo significant functional changes at around 280 days or so of culture. If that number strikes you as nonrandom, you’re onto something. It marks the time from a child’s conception to birth. “It’s as if they’ve got a clock,” Pasca said.
In 2015, Pasca (who by then had his own lab at Stanford Medicine) and his group published a paper in Nature Methods describing the transformation of iPS cells into neural organoids. Floating in laboratory glassware, these balls consisted of cells that were organized in much the same way cells are organized in the cerebral cortex.
“When Sergiu showed us his results, we were really blown off our chairs.”
Jan Carette, PhD, an associate professor of microbiology and immunology
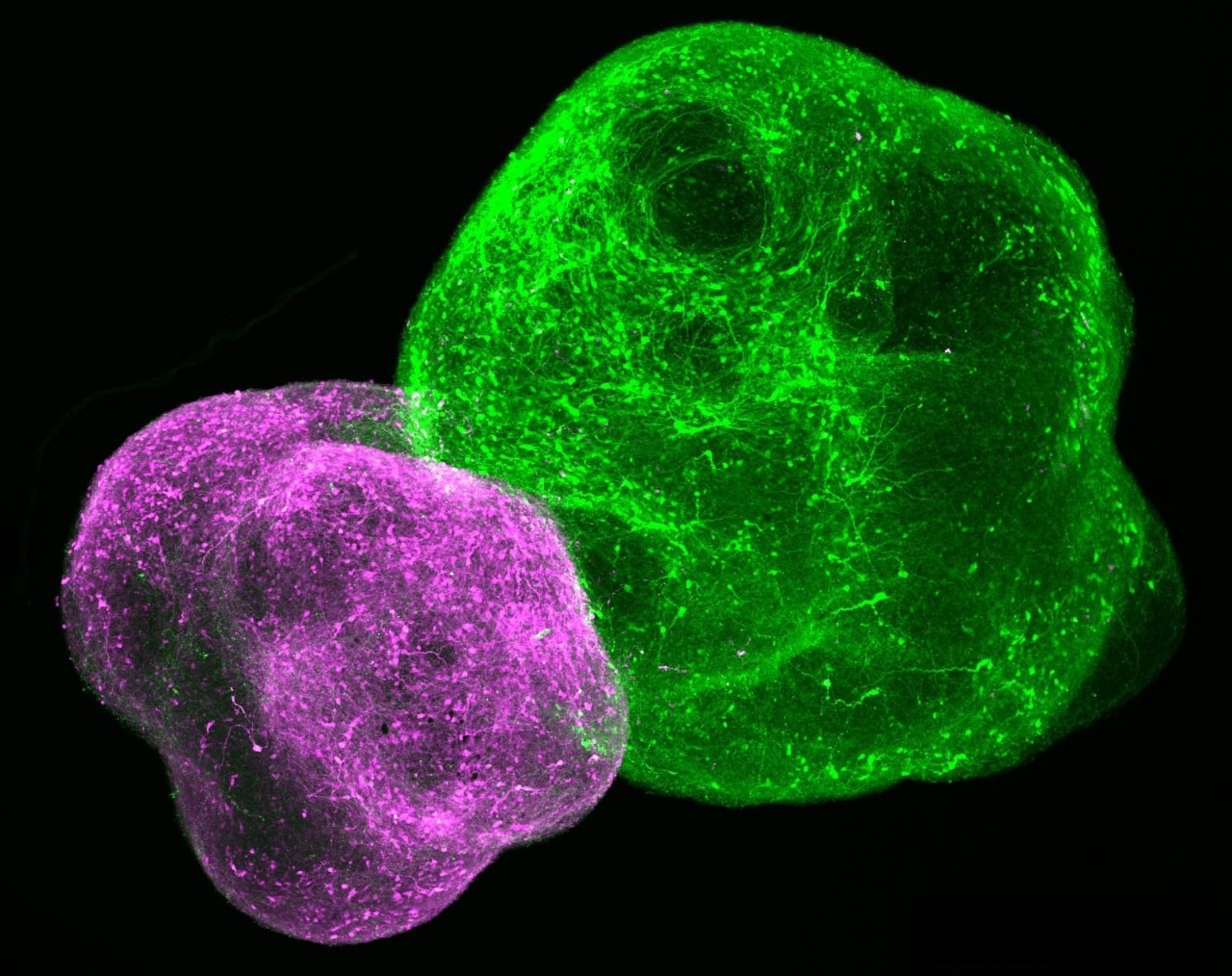
In a 2017 paper in Nature, Pasca’s team showed how they’d created two distinct types of organoids — one representing the cortex, the other the subpallium, which is a brain region beneath the cortex that plays a significant role in fetal development and then more or less fades away.
The researchers let the two organoids cuddle in the same dish, gave them time to fuse together (they do this on their own!), and watched a set of nerve cells called interneurons migrate from the subpallium into the adjacent cortex.
There, the interneurons made contact with the very neurons they’re fated to meet in real-world fetal development and set up shop, sprouting branching brush-like tails so they could receive input from one another and forming working relationships with neurons in the cortex to generate neural circuits.
Thus began the era of the assembloid: the union of two organoids to permit the observation of different parts of the brain (or, indeed, other organs) connecting and communicating as they do in a living nervous system. Pasca began to use assembloids as workhorses for learning more about the brain.
In the Nature paper, Pasca’s team showed that interneurons’ migration to the cortex proceeds in discrete, stuttering jumps. But in assembloids derived from Timothy syndrome patients, the jumping of interneurons originating from the subpallium is impaired — they jump more often, but the jumps are shorter — with the net result being that they fail to integrate into the appropriate circuitry in the cortex.
By 2020 he’d shown, in a paper published in Cell, how to string together three organoids — representing skeletal-muscle tissue, the spinal cord and the region of the cerebral cortex that’s responsible for voluntary movement — into an assembloid mimicking the neuromuscular apparatus that controls every voluntary move we make. Stimulating the cortex organoid on one end of the three-part assembloid caused the muscle mass at the other end to twitch.

Paralysis in a dish
“When Sergiu showed us his results, we were really blown off our chairs,” said Jan Carette, PhD, associate professor of microbiology and immunology, whose team is collaborating with Pasca’s group to unravel the pathology of crippling viruses similar to the one that causes polio.
Carette is a virologist who focuses on enteroviruses, a group of viruses so named because they often infect the intestine (“entero” is the Latin root of the term “entrails”) — typically without causing much trouble. It’s when these viruses break out and hole up in other thoroughfares and orifices that symptoms develop.
On rare occasions, enteroviruses get into the nervous system. Then symptoms can be severe. The once-dreaded poliovirus, responsible for the crippling childhood disease that is polio, is an enterovirus. Fortunately, we don’t see many cases of polio anymore — it’s been all but eradicated globally, thanks to effective vaccination drives.
But a couple of poliovirus’s close cousins — notably one called EV-D68 — worry those who pay attention to emerging diseases. Unlike most enteroviruses, EV-D68 doesn’t replicate well in the intestinal tract.
Instead, it’s well adapted to our upper respiratory tracts. Like other respiratory viruses, it’s transmitted by coughing, sneezing and sometimes even by simply breathing. If it infects the spinal cord, EV-D68 can cause a paralytic syndrome called acute flaccid myelitis.
“Viral replication seems to happen mainly in the spinal cord. That’s a difficult place to get tissue from.”
Carette, whose team is collaborating with Pasca’s group to unravel the pathology of crippling viruses similar to the one that causes polio
Cases of the syndrome are still rare but have recurred in alternate years with increasing frequency since 2014. (An exception is 2020 — cases dipped, probably a result of COVID-19-pandemic-induced social distancing, Carette said.) In all, there have been about 700 cases of confirmed acute flaccid myelitis in the United States since August 2014, when the Centers for Disease Control and Prevention began tracking them.
“We’re interested in understanding how the virus causes paralysis,” Carette said. “Somehow, the virus reaches the spinal cord, invades motor neurons and damages them so they can’t direct normal muscle movement. We’re asking how, exactly, these viruses interact with human cells. Which cells do they infect? What receptor or receptors on those cells do they hook up with in order to do this? How do they then cause damage to these cells? And how does the immune system respond?”
Questions like those are more easily asked than answered. EV-D68 is tough to study because mice, the workhorses of biomedical research, make lousy models for enteroviral disease. Like poliovirus, EV-D68 naturally infects only primates. That’s great in the sense that, in the absence of nonhuman reservoirs, it may be possible in principle to eradicate the virus by vaccinating everybody, should a vaccine be developed.
But from a researcher’s standpoint, it’s a challenge. And not the only one. “Viral replication seems to happen mainly in the spinal cord,” Carette said. “That’s a difficult place to get tissue from.”
So Carette is collaborating with Pasca, using the latter’s assembloid construct to induce what one could call “paralysis in a dish.” “We’re looking hard at the spinal-cord/muscle junction, where infection probably occurs,” Carette said. “There’s a variety of neuronal cell types in the spinal cord. Nobody knows, yet, which of them is the virus’s target.”

By introducing the virus to spinal-cord organoids or spinal-cord/skeletal-muscle assembloids, then inspecting individual cells to see which ones now contain EV-D68 genetic material revealing the virus’s presence, the scientists hope to determine which types of neurons are successfully penetrated by EV-D68. They also want to learn how the virus gets in.
Monitoring the activation levels of essentially all the genes inside an infected neuron may reveal which genes’ activity levels jump or shrink most markedly in response to infection. That will provide clues to how infected cells attempt to stave off viral replication and to summon assistance from the immune system.
Carette also hopes to use assembloids to compare the virulence of EV-D68 strains from 2014 with those from later years to see if the virus is getting more virulent and, if so, why.
“We’re very excited about this project,” he said. “The options are limitless.”
The developing brain
Anca Pasca, MD, an assistant professor of pediatrics, was a key, hands-on co-author of Sergiu Pasca’s 2015 Nature Methods study describing the first generation of cortex-mimicking organoids. Now she’s focusing on the effects of hypoxia — too little oxygen — on the developing fetal brain, exploring its effects on the interneurons that migrate to the cerebral cortex from the fetal subpallium during pregnancy.
Once inside the cortex, these interneurons form complex circuits with resident excitatory neurons, shaping the bursts of activity those neurons produce and discouraging bouts of frenzied firing that characterize neurodevelopmental disorders from epilepsy to autism to schizophrenia.
A deficit of interneuron hookups puts the developing brain at risk for these conditions, Anca Pasca said. And autopsies have revealed that the cerebral cortex of a prematurely delivered baby contains reduced numbers of interneurons compared with that of a full-term newborn.
“We thought this might be related in part to hypoxia,” she said. “Even before birth, fetuses that have been deprived of oxygen due to congenital heart disease have smaller brains. And prematurely born babies’ lungs are immature. We have to breathe for them, which we do by applying oxygen-supplying masks to their noses or putting breathing tubes into their windpipes. But because their lungs are undeveloped, the oxygen doesn’t easily pass from the lungs into the blood.”
“We can see that the cells are already actually trying to make this protein. Can we help them out by giving them more of it?”
Anca Pasca, MD, an assistant professor of pediatrics
Is hypoxia killing interneurons before they can embark on their journey to the cortex? Preventing that migration from getting underway? Slowing it to a crawl during a critical period, so the interneurons never quite reach their destination? Or preventing hookups once the interneurons arrive?
To find out, Pasca’s been working with a cortex/subpallium assembloid, like the ones Sergiu Pasca’s team developed in 2017. By exposing these assembloids to hypoxia, she has shown that one of the major problems caused by hypoxic conditions lies in impaired migration of interneurons on their way to the cortex.
“They get stuck on the road,” she said. “They don’t migrate as well. So, fewer of them later integrate into the circuitry in the cortex.”
Her team has identified a key substance, produced naturally in the brain, that counters this impairment and that, used as a supplement, could possibly ameliorate the damage induced by hypoxia.
“We can see that the cells are already actually trying to make this protein,” Pasca said. “Can we help them out by giving them more of it?” She hopes to move rapidly into experiments with living nonhuman primates to clear the path for a clinical trial.
What comes next?
In the cards are more-ambitious assembloids. Sergiu Pasca is experimenting with a four-organoid version that starts by hooking the cortex to a brain region called the striatum, which plays key roles in both physical movement and psychological motivation, the two driving forces of motion.
That opens the door to more-intensive study of movement and psychiatric disorders. Pasca and his colleagues published the entire recipe for generating cortex/striatum assembloids from scratch in Nature Biotechnology in 2020. He also intends to fold in another brain region called the thalamus, which feeds sensory input from the outside world to the cortex.
“I don’t think we’ve reached the limits of complexity yet, but we will,” he said. “We can in principle put four, five, maybe six organoids together before it becomes too challenging for nutrients and growth factors in the culture broth to reach cells deep within these multi-cellular structures.”
In any case, organoids in a dish don’t contain every cell type resident in the tissues they mimic. They lack blood vessels, for example, whose absence means an assembloid can absorb oxygen and nutrients only at its surface. That’s growth-limiting.
One way to get around such limitations is through transplantation. Pasca’s team has been grafting human cortex organoids into the brains of living laboratory rats.
Preliminary indications are that the human brain tissue not only engrafts into the rats’ brains but also receives sensory input and becomes permeated by blood vessels supplying oxygen and nutrients and carting away metabolic waste, in turn permitting the engrafted circuitry to keep growing larger, better integrated, more complex …
And … more human?
That’s a good question, said Pasca. As are these: What animals should be considered off-limits for human brain-tissue implants? (All primates, Pasca answers.) Is it acceptable to provide sensory input to a human assembloid, whether in a dish or in a living animal? What about introducing a pain pathway?
Cutting to the chase: Could assembloids ever acquire anything approaching consciousness? The short answer is that it’s a long way off, and it may never be possible. Still, this is an example of how Pasca’s work defines new frontiers of biomedical ethics as it helps define the foundations of neurodevelopment.
“One in 5 people have some kind of psychiatric illness. But the brain is hard to study, and the success rate for finding new drugs for brain disorders is lower than for any other organ — largely because, until now, we couldn’t study live human brain tissue from patients.”
Sergiu Pasca, MD, an associate professor of psychiatry and behavioral sciences
Careful about crossing any ethical lines, Pasca has been proactive all along, consulting at every step of the way with Stanford bioethicists; participating in the National Institutes of Health–sponsored Brainstorm Project, designed to develop ethical guidelines for organoid- and assembloid-associated research; and engaging in discussions with a committee convened by the National Academies of Science, Engineering and Medicine to probe the ethics of neural organoids, transplants and mixed-species experimentation.
Meanwhile, the rats into whose brains Pasca and his colleagues have transplanted lab-grown human cortex organoids don’t seem to be acting like anything but the rats they still are.
“We put them through a battery of behavioral tests but found no discernible movement or memory deficits or brain-wave abnormalities,” Pasca said. Nor did the scientists observe any signs of enhanced performance in these murine organoid-recipients.
There’s always been a market for depicting the future as a scary scenario. But nothing like a living brain inhabiting a dish is in the forecast. Meanwhile, back in the here and now, the real danger may lie not in assembloid research’s moving too quickly, but in its proceeding too slowly. First and foremost, Pasca told his TED audience, “an assembloid is not a brain — it’s just a working model of some of the circuitry resident in a living, functioning human brain.”
And second, he told them, “1 in 5 people have some kind of psychiatric illness. But the brain is hard to study, and the success rate for finding new drugs for brain disorders is lower than for any other organ — largely because, until now, we couldn’t study live human brain tissue from patients.”
Now we can.
At the end of his talk, Pasca got a standing ovation.